The Use of Concentrated Growth Factors and Autogenous Bone for Periodontal Soft Tissue Augmentation and Bone Regeneration: A Case Report
Xuan Ying, Yue Chen*, Ning Luo, Lei Shi, Yuxin Tong, Jiayu Zhang
Department of Periodontology, College of Stomatology, Xi’an Jiaotong University. Xi’an, Shaanxi, People’s Republic of China.
- Corresponding Author:
- Tel: 86-15224023939
E-mail: chenyue0306@126.com
Received date: June 20, 2017; Accepted date: July 11, 2017; Published date: July 18, 2017
Citation: Ying X, Chen Y, Luo N, et al. The Use of Concentrated Growth Factors and Autogenous Bone for Periodontal Soft Tissue Augmentation and Bone Regeneration: A Case Report. Electronic J Biol, 13:3
Abstract
Objective: To achieve periodontal soft tissue and bone augmentation using concentrated growth factors (CGFs) and autogenous bone in a single operation. Methods: A patient who presented with a thin gingival biotype and severe labial alveolar bone defect of the mandibular anterior teeth required orthognathic surgery. CGF and autogenous bone were used to achieve periodontal soft tissue and bone augmentation during the orthognathic surgery. Photos of the operative area were taken preoperatively and 12 days, 1, 3, 6 and 12 months after surgery to evaluate the esthetic effect of periodontal soft tissue augmentation. The operative area was scanned using cone-beam computed tomography, in order to measure augmentation of the alveolar bone before and at 3, 6 and 12 months after surgery. Results: CGF treatment resulted in marked vascularization on the 12th day after the surgery. During the 12 months clinical follow-up, intraoral examinations showed that the periodontal soft tissue maintained complete root coverage and the increased gingival thickness remained stable. The color, shape and texture of CGF-treated region gradually became similar to that of the surrounding gingiva, achieving a satisfying esthetic effect. Moreover, cone-beam computed tomography showed that the labial alveolar bone of the mandibular anterior teeth was also augmented in height and thickness, which gradually stabilized in the 6th month after surgery. Conclusion: CGF combined with autogenous bone can achieve periodontal soft tissue and bone augmentation. A 12 months follow-up period revealed stable augmentation and a satisfying esthetic effect.
Keywords
Autogenous; Bone augmentation; Concentrated growth factors; Periodontal soft tissue; Orthodontics; Orthognathics.
1. Introduction
During orthodontic treatment, the movement of teeth always results in some problems, such as gingival recession, absorption of the labial alveolar bone and dehiscence, particularly in the area of mandibular anterior teeth with its thin layer of soft tissue and poor blood supply, which is provided by small peripheral vessels. Moreover, the lack of soft tissue and bone defects typically co-exist in mandibular anterior teeth. Such patients typically require a staged operation to achieve soft tissue augmentation as well as bone regeneration. Soft tissue augmentation can benefit for bone regeneration. However, a staged operation prolongs the treatment period, cost, and the discomfort of patients. Here, we report the case of an orthodontic patient, who presented with a thin gingival biotype and a severe labial alveolar bone defect of the mandibular anterior teeth, requiring orthognathic surgery. Concentrated growth factors (CGFs) are the third generation of blood extracts which contains a markedly higher concentration of fibrinogen, coagulation factors and CD34+ cells. We used CGF and autogenous bone to achieve periodontal soft tissue and bone augmentation. Follow-up over a period of 12 months demonstrated stable augmentation and a pleasing esthetic effect.
2. Case Report
2.1 Case presentation
In May 2014, a 30 years old female patient was referred to the Department of Orthodontics, College of Stomatology, Xi’an Jiaotong University, Xi’an, China, with the chief complaint of a mandibular protrusion that had persisted for more than 20 years. She had accepted a plan of orthognatic-orthodontic treatment. Pre-operative orthodontic alignment was achieved over a period of 10 months (June 2014 to April 2015) on the basis of tooth #25 extraction. Second-degree mobility was found in teeth #33−43 during the orthodontic treatment process. And in April 2015, her doctor referred her to the Department of Periodontics for an excessively thin labial alveolar bone of the mandibular anterior teeth. The patient was in a good general condition and had no history of smoking.
2.2 Clinical examination
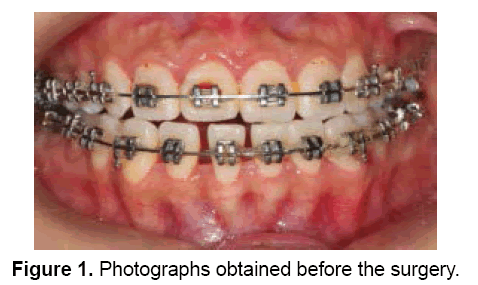
During the intraoral examination, DI (1), CI (1), the labial gingiva at teeth #33-43 were noted to be of a thin gingival biotype, with a pink color and tough texture. Periodontal clinical measurements of teeth #33-43 included determination of BOP (which was negative), probing depth (1-2 mm), subgingival calculus (which was negative), recession depth (0 mm), and width of the attached gingiva (2-3.5 mm), using the Williams periodontal probe. There was second-degree mobility in teeth #33−43 (Figure 1). Cone-beam computed tomography (CBCT) showed that the labial alveolar bone of the mandibular anterior teeth had been absorbed to one-third of the apical area. Blood analysis was unremarkable.
2.3 Diagnosis and treatment planning
A diagnosis of labial dehiscence or fenestration of teeth #33-43 was made. Treatment planning involved initial periodontal therapy, followed by orthodontic, orthognatic and periodontic treatment. This was followed by periodontal surgery to achieve soft tissue and bone augmentation during the orthognathic surgery.
2.4 Presurgical preparation
The procedure was described to the patient and written informed consent was obtained. Oral hygiene instructions with emphasis on brushing habits were given. Supragingival scaling was performed. After the initial periodontal therapy, the patient was periodically recalled in order to ensure that the periodontal tissue was not inflamed.
2.5 Surgical treatment
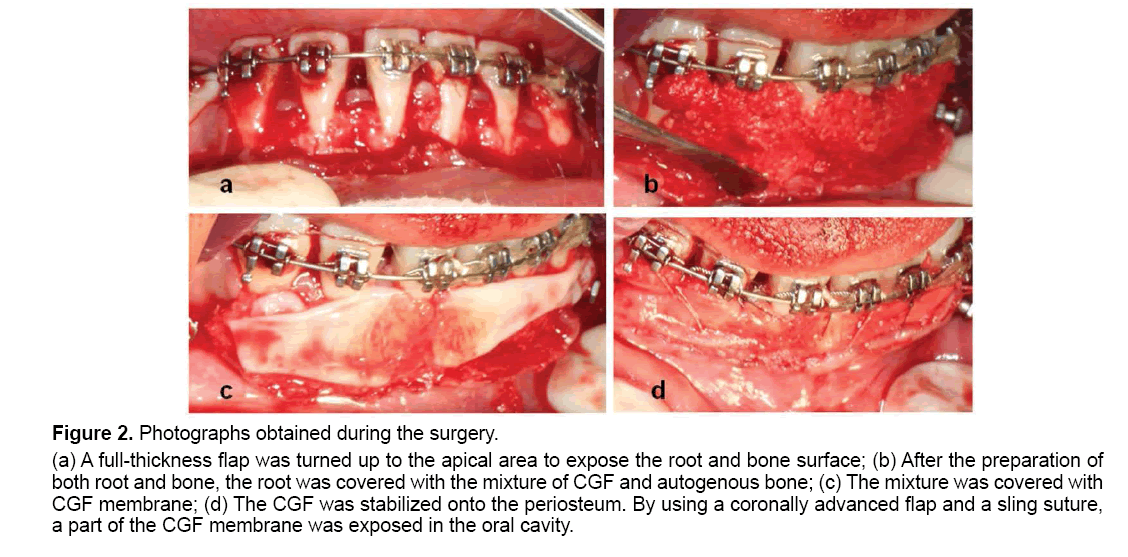
The orthognathic surgery and the periodontal surgery were performed under general anesthesia on May 13, 2015. During the orthognathic surgery, a horizontal incision was made approximately 2 mm apical to the labial gingival margin of teeth #33-43 and a full-thickness flap was then turned up to the apical area to expose the root and bone surface. The root prominence was reduced and the root surface was modified with EDTA. The alveolar bone preparation was obtained. Four tubes (9 ml each) of venous blood were obtained from the patient by using a Vacuette needle and a vacuumpacked Vacuette. The blood was transferred into a test tube and centrifuged in a Medifuge (Thermo Fisher Scientific, Waltham, MA, USA) for the preparation of CGF. After 13 min of centrifugation, the whole blood was divided into four layers, i.e., the serum layer, the buffy coat layer, the CGF+ stem cell layer and the red cell layer from top to bottom of the tube. The serum layer and a part of the red cell layer were discarded, while the buffy coat layer, the CGF+ stem cell layer and about 1 cm of the red cell layer were retained. The CGF layer was collected and placed into a mold to produce four CGF membranes. Meanwhile, the autologous bone (cancellous bone and a small amount of cortical bone removed from mandible during orthognathic surgery) was ground to a powder and mixed with a small portion of CGF that had been cut into pieces of approximately 1 mm × 1 mm in size. The root was covered with the mixture and the remaining CGF membranes were placed over it. The CGF was stabilized onto the periosteum. By using a coronally advanced flap and a sling suture, a part of the CGF membrane was exposed in the oral cavity (Figure 2).
Figure 2: Photographs obtained during the surgery.
(a) A full-thickness flap was turned up to the apical area to expose the root and bone surface; (b) After the preparation of
both root and bone, the root was covered with the mixture of CGF and autogenous bone; (c) The mixture was covered with
CGF membrane; (d) The CGF was stabilized onto the periosteum. By using a coronally advanced flap and a sling suture,
a part of the CGF membrane was exposed in the oral cavity.
On the second day after surgery, the patient was equipped with a final splint with intermaxillary elastics, which was worn for 4 weeks. In the 4th week, the splint was removed. After surgery, the patient was instructed to avoid mechanical trauma and thus, abstain from chewing, brushing, and flossing around the surgical area for 4 weeks. Plaque control in the surgically treated area was maintained by chlorhexidine (0.12%) mouthwash for 4 weeks. The patient was prescribed 600 mg ibuprofen as needed.
2.6 Follow-up visit
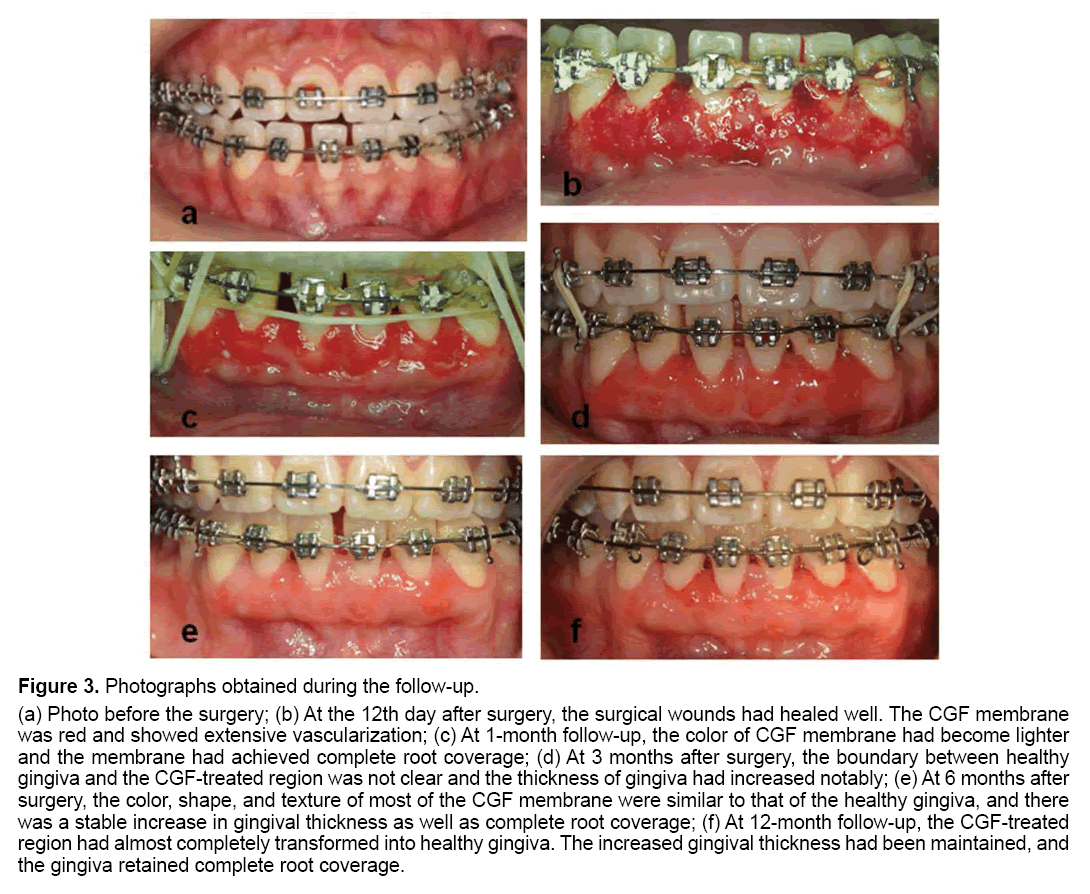
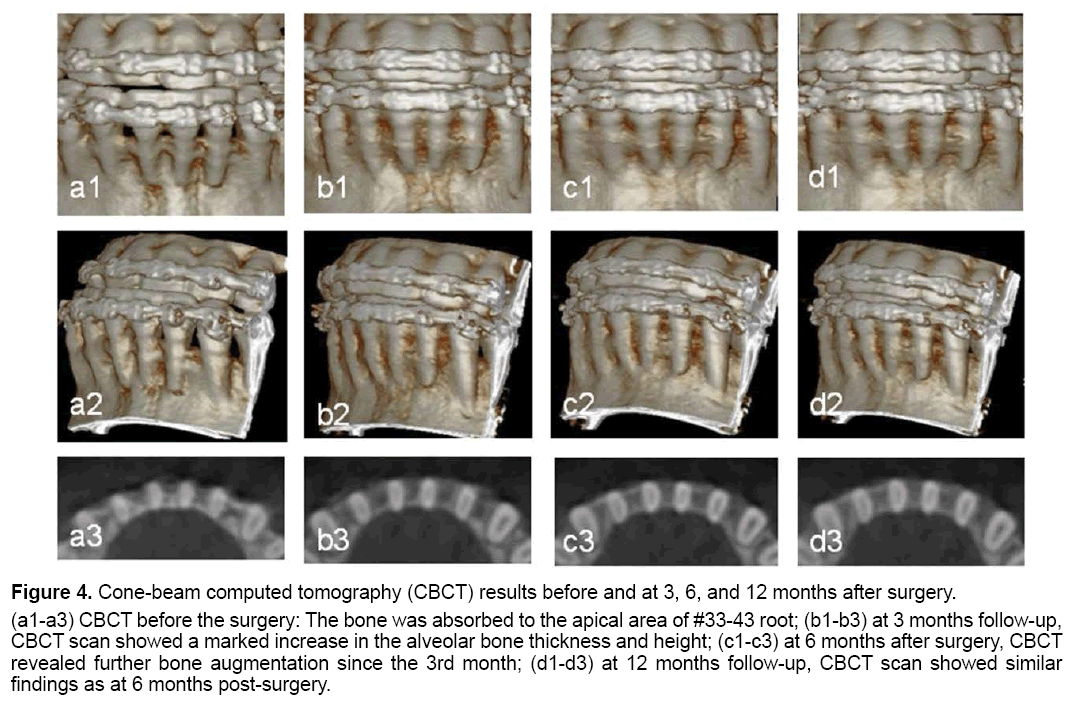
The sutures were removed on the 12th day after surgery and the surgical wounds were healed well. The CGF membrane was red and showed extensive vascularization. At 1 month follow-up, the patient had maintained good oral hygiene and the gingival edema were significantly reduced. The color of the CGF membrane had become lighter and achieved complete root coverage. Three months after surgery, the boundary between healthy gingiva and the CGFtreated region was not clear and thickness of the gingiva had increased notably. CBCT at this time showed an increase in alveolar bone thickness and height. At 6 months after surgery, the color, shape and texture of most of the CGF membrane were similar to that of the healthy gingiva, and there was a stable increase in the gingival thickness as well as complete root coverage. CBCT also revealed further bone augmentation since the 3rd month. The CGF-treated region had almost completely transformed into healthy gingiva at the 12-month follow-up. The increased gingival thickness was maintained, and the gingiva retained complete root coverage. The gingival margin was slightly swollen, as the patient’s oral hygiene was not optimal. CBCT showed similar findings as at 6 months post-surgery (Figures 3 and 4).
Figure 3: Photographs obtained during the follow-up.
(a) Photo before the surgery; (b) At the 12th day after surgery, the surgical wounds had healed well. The CGF membrane
was red and showed extensive vascularization; (c) At 1-month follow-up, the color of CGF membrane had become lighter
and the membrane had achieved complete root coverage; (d) At 3 months after surgery, the boundary between healthy
gingiva and the CGF-treated region was not clear and the thickness of gingiva had increased notably; (e) At 6 months after
surgery, the color, shape, and texture of most of the CGF membrane were similar to that of the healthy gingiva, and there
was a stable increase in gingival thickness as well as complete root coverage; (f) At 12-month follow-up, the CGF-treated
region had almost completely transformed into healthy gingiva. The increased gingival thickness had been maintained, and
the gingiva retained complete root coverage.
Figure 4: Cone-beam computed tomography (CBCT) results before and at 3, 6, and 12 months after surgery.
(a1-a3) CBCT before the surgery: The bone was absorbed to the apical area of #33-43 root; (b1-b3) at 3 months follow-up,
CBCT scan showed a marked increase in the alveolar bone thickness and height; (c1-c3) at 6 months after surgery, CBCT
revealed further bone augmentation since the 3rd month; (d1-d3) at 12 months follow-up, CBCT scan showed similar
findings as at 6 months post-surgery.
3. Discussion
Dehiscence may be caused by many reasons, including local anatomic characteristics, such as tooth buccoversion, thin labial alveolar bone, and large tooth protuberance. Other reasons, such as occlusal trauma, periodontitis and orthodontic treatment can also lead to dehiscence. It is quite common for dehiscence to appear during orthodontic treatment, and this may lead to adverse effects, such as the disappearance or decrease of orthodontic anchorage, gingival recession, root exposure and loosening or even loss of a tooth [1]. Guided bone regeneration must be performed to resolve dehiscence. The thin soft tissue and poor blood supply further complicates achieving a good and stable curative effect after periodontal regenerative surgery. Lee et al. [2] have reported that the thin gingival biotype is frequent in Asians.
Both free gingival flap (FGG) and connective tissue flap (CTG) operations require an additional surgical area, which may cause more discomfort during or after surgery. A large amount of connective tissue is needed to cover multiple adjacent defects. In some cases, it may be difficult to harvest sufficient CTG or FGG in the presence of a flat palatal profile or if the masticatory mucosa is too thin [3]. In the current patient with a thin gingival biotype and a severe labial alveolar bone defect of the mandibular anterior teeth, we therefore opted to use CGF and autogenous bone.
The use of CGF was first proposed by Saccol et al. in 2006 [4]. It is the third generation of blood extracts after platelet-rich plasma and platelet-rich fibrin. CGF contains a markedly higher concentration of fibrinogen, coagulation factors and CD34+ cells than the aforementioned preparations and is considered as a new type of biological material [5].
CGF improves the thickness and promotes healing of soft tissue; however, it has not been reported to date that CGF can be used with partial exposure in the oral cavity to induce changes in the gingiva. We propose that the mechanism involves high quantities of growth factors in the platelets, which can stimulate cell proliferation, matrix remodeling and angiogenesis, and which are released by centrifugation [6]. Vascular endothelial growth factor (VEGF) is a key regulator of physiological angiogenesis, which represents a critical step in this remodeling. It has the ability to induce vascular leakage, promote endothelial cell proliferation, and initiate vascular branch formation [7]. VEGF functions to induce vascularization of the CGF membrane. EGF stimulates epidermal growth and keratinization, but also promotes angiogenesis in the CGF membrane and thus, CGF causes changes in the gingiva [8]. Moreover, CD34+ cells have therapeutic potential in terms of both vasculogenesis and osteogenesis [9]. CD34+ cells are capable of inducing neovascularization of the CGF membrane. In addition, CGF ensures sealing of soft tissue defects, which enhances the effect of guided bone regeneration by promoting healing. Moreover, the three-dimensional CGF membrane network structure contains a high concentration of platelets and growth factors, which can be released and act effectively over a prolonged period.
It is known that the best bone grafts have three basic characteristics: osteoconduction, osteoinduction, and osteogenesis [10]. The bone graft materials used in bone regeneration at present include autologous bone; allogenous bone grafts, bone xenografts and synthetic grafts. Autologous bone is still considered the clinical “gold standard” for bone regeneration, as it has all three characteristics without any associated immune response [11]. In this case, simultaneous periodontal and orthognathic surgery enabled us to obtain sufficient autologous bone. CBCT at the 3 months visit showed a notable increase in alveolar bone thickness and height, with a rough surface. At 6 months after surgery, surface of the alveolar bone became smooth, although little bone regeneration had occurred. CBCT at 12 months after surgery was similar to the former.
In the current case, we observed that most of the bone gain was concentrated in the interproximal bone thickness, rather than labial bone thickness. The gain in labial alveolar bone thickness was limited. We speculate that this is related to fast absorption of the autogenous bone. If we add a certain percentage of artificial bone material to autogenous bone, allowing the new bone structure to stay intact longer, a better osteogenesis effect may be achieved.
Additionally, postoperative care is also important for a curative effect. The intermaxillary traction for 4 weeks post-operatively, guaranteed stability around the surgical area, ensuring that the CGF and autogenous bone could attach to the root surface closely. Subsequent timely visits for periodontal maintenance therapy ensured cleanliness and plaque control of the surgical area, which improved prognosis.
4. Conclusion
Here we report the case of an orthodontic patient with a thin gingival biotype and severe labial alveolar bone defect of the mandibular anterior teeth who required orthognathic surgery. CGF and autogenous bone were used to achieve periodontal soft tissue and bone augmentation during the orthognathic surgery. After 12 months, the color, shape and texture of the CGF membrane, which was partly exposed in the oral cavity, gradually became similar to the healthy gingiva, owing to its vascularization. The thickness of the gingiva increased notably and complete root coverage was retained. The effect of soft tissue augmentation was esthetically satisfying and stable. CBCT showed that the labial alveolar bone of the mandibular anterior teeth was augmented in height and thickness, and gradually stabilized over 6 months after surgery. It is reasonable to speculate that the curative effect in this case was closely related to the use of CGF and autogenous bone, as well as effective postoperative care. This case implies that patients presenting with thin labial alveolar bone and a thin gingival biotype can achieve periodontal soft tissue and bone augmentation by using CGF and autogenous bone in a single rather than staged operation. This is important for broadening the spectrum of adaptation and reducing the complications of orthodontic treatment. It can also be used to improve the periodontal soft tissue and bone defects in patients with periodontitis. Further studies, extending beyond 12 months should be performed to investigate the long-term effects of this approach.
References
- Min L, Li Z, Yanmin W. (2013). Present research situation of the orthodontic related alveolar bone defects. Int J Stomatol. 40: 500-502.
- Lee SA, Kim AC, Prusa Jr LA, et al. (2013). Characterization of dental anatomy and gingival biotype in Asian populations. CDA J. 41: 30-39.
- Rotundo R, Pini-Prato G. (2012). Use of a new collagen matrix (mucograft) for the treatment of multiple gingival recessions: Case reports. Int J Periodontics Restorative Dent. 32: 413-419.
- Rodella LF, Favero G, Boninsegna R, et al. (2011). Growth factors, CD34 positive cells and fibrin network analysis in concentrated growth factors fraction. Microsc Res Tech. 74: 772-777.
- Doğan ŞB, Dede FÖ, Ball U, et al. (2015) Concentrated growth factor in the treatment of adjacent multiple gingival recessions: a split-mouth randomized clinical trial. J Clin Periodontol. 42: 868-875.
- Giuseppe Intini. (2009). The use of platelet-rich plasma in bone reconstruction therapy. Biomater. 30: 4956-4966.
- Ferrara N, Gerber H, LeCouter J. (2003). The biology of VEGF and its receptors. Nat Med. 9: 669-676.
- Chim SM, Tickner J, Chow ST, et al. (2013). Angiogenic factors in bone local environment. Cytokine Growth Factor Rev. 24: 297-310.
- Guanghui Li, Xi Wang, Jian Cao, et al. (2014). Coculture of peripheral blood CD34+ cell and mesenchymal stem cell sheets increase the formation of bone in calvarial critical-size defects in rabbits. Br J Oral Maxillofac Surg. 52: 134-139.
- Cypher TJ, Grossman JP. (1996). Biological Principles of Bone Graft Healing. J Foot Ankle Surg. 35: 413-417.
- García-Gareta E, Coathup MJ, Blunn GW. (2015). Osteoinduction of bone grafting materials for bone repair and regeneration. Bone. 81: 112-121.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences