Potent in vitro Anti-tumor Activity of Symplocos racemosa against Leukaemia and Cervical Cancer
Bhuvan P. Raval, Maulik P. Suthar, Rakesh K. Patel
1K.J. College of pharmacy,Vadasma,north Gujarat,India
2S.K. Patel College of pharma Edu and Res,ganapat University,At: Kherva,north Gujarat,India
- Corresponding Author:
- Bhuvan P. Raval
K.J. College of pharmacy
Vadasma,north Gujarat,India
Tel & Fax: +91 (0)2762286324
E-mail: bhuvan_raval2002@yahoo.co.in
Abstract
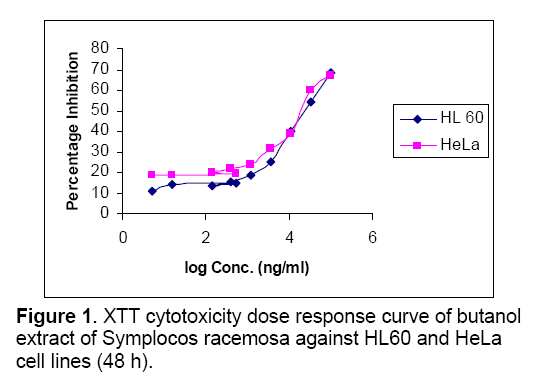
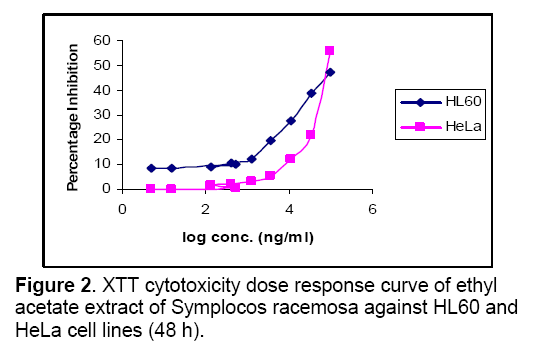
Objective: Assessment of anti-tumor activity of Symplocos racemosa Roxb. (Symplococaceae) plant extracts. Methods: Plant bark was procured, identified and verified. Chloroform, butanol and ethyl acetate extracts were prepared and their cytotoxic activity determined using the XTT salt based cytotoxicity assay in 96- micro plate format against one leukaemia and one cervical cancer cell line. Cyclophosphamide was used as positive control. Absorbance was measured after 48 h at 450 nm. Results: Total 3 extracts evaluated, butanolic and ethyl acetate extracts demonstrated strong cytotoxic potential overall. Cell proliferation assay showed dose depended inhibition of cell growth. Butanolic extract was found cytotoxic against HL 60(Human leukemia cell line, IC50= 27183 ng/ml), HeLa (Human cervix cancer cell line, IC50= 22861 ng/ml), Ethyl acetate extract was found to be less cytotoxic against HL 60 (IC50= 117084 ng/ml), HeLa (IC50= 137151 ng/ml). Chloroform extract displayed no cytotoxicity against both cell lines. Conclusion: S.racemosa shows potential anti-tumor activity against leukaemia and cervical cancer.
Keywords
HL60; HeLa; cell line; plant based drug.
1. Introduction
Cancer is the second leading cause of death in the world [1]. Throughout history and across the world,the plant kingdom has provided a variety of medicines for cancer treatment. In modern times,plants have been a source of analgesics,anti-inflammatories,antiasthmatics,antiarrhythmic agents,antihypertensives,antimicrobial agents known to be numerous. Currently over 60% of the drugs are derived in one or other way from natural source including plant,marine organism and micro-organism [2]. There are worldwide efforts to discover anticancer agents from plants.
Symplocos racemosa is a traditional Ayurvedic medicine. The plant,plant extract and isolated salireposides have been extensively investigated in several laboratories for their menstrual regulatory effects and a number of reports are available confirming their action. In addition,researchers have evaluated the anti-bacterial effect of S. racemosa extracts against acne inducing bacteria [3].
Symplocos racemosa is proven to contain the dithiadiazetidin namely symploate,as well as linoleic acid,oleic acid,salireposide,symplocososide,beta-sito-glycoside,symponoside,symplososide,symploveroside,benzoylsalireposide and salireposide [4-6]. No cytotoxicity of S. racemosa has been reported.
2. Methods
2.1 Plant Collection
Ethnobotanical literature was reviewed and plant bark was procured from Lallubhai Vrajlal Gandhi,Ahmedabad,Gujarat,India and authenticated by Dr. Ritesh Vaidya,Bioscience Department,Ganpat University.
2.2 Aqueous extract preparation
Bark was shed,dried and ground by simple grinding machine to visually fine powder. No particle size analysis was done. The fine powder of S. racemosa (500g) was exhaustively extracted with 1.5l methanol for 24h at room temperature. After 24h,the methanol was evaporated and dried residue was subjected to successive partition with chloroform,butanol and ethyl acetate respectively. All three layers were collected individually and were subjected to evaporation and drying. Extracts were dissolved in 2% Dimethyl Sulphoxide (DMSO) solution. 10 serial dilutions were prepared from each of the extract ranging from 100μg/ml to 0.005μg/ml.
2.3 Preparation of media DMEM (1X Liquid Medium with FBS & antibiotic)
5% less distilled water was measured out than desired total volume of medium. Powdered medium was added to water with gentle stirring. 3.7g of NaHCO3 was added per liter of medium. Diluted to a desired volume with water and stirred until dissolved. pH of medium was adjusted to 0.2-0.3 below desired final working pH. After pH adjustment,was kept in a closed container,Sterilized immediately by membrane filtration. DMEM,Antibiotic,and FBS (Fetal bovine Serum) were prepared.10% FBS and 5 ml antibiotics were added into the DMEM [7].
2.4 Subculture of cell lines
Cultures were viewed using an inverted phase contrast microscope. Centrifugation for subculture was not performed unless the pH of the medium was acidic which indicated the cells overgrown and were not recovered. Cells were centrifuged at 150g for 5 minutes,re-seeded at a slightly higher cell density and 10- 20% of conditioned medium added to the fresh media. Small sample of the cells from the cell suspension was taken. Cells/ml was calculated and the desired number of cells re-seeded into freshly prepared flasks without centrifugation just by diluting the cells [7].
2.5 XTT assay
XTT assay was employed to assess cell proliferation. Viable cells were seeded into 96-well microtitre plates at 5×104 cells/well in DMEM (Dulbecco’s modified Eagle’s medium) supplemented with FBS (fetal bovine serum),100units/ml penicillin,100 μg/ml streptomycin and cultured in a humidified atmosphere of 5% CO2 and 95 air at 370C. 180 μl of cell suspension was cultured with 20μl of various concentrations of all three extracts (0.005-100μg/ml) dissolved in 2% DMSO solution. Control cells were incubated in culture medium only. Wells containing only media were considered as a blank. All extract dilution doses were tested in duplicates.
The cell proliferation is based on the ability of the mitochondrial succinate-terazolium reductase system to convert yellow tetrazolium salt XTT (sodium 3´-[1- (phenylaminocarbonyl)- 3,4- tetrazolium]-bis (4 methoxy- 6-nitro) benzene sulfonic acid hydrate) to orange formazan dye. The test denotes the survival cells after toxic exposure. 50μl of XTT mixture was added to each well. After 48h incubation at 370C temperature and 5% CO2,the absorbance of soluble formazan product produced by viable cells was measured at 450nm using ELISA plate reader (Thermo,USA). Reference wavelength used was 650 nm.Percentage inhibition of the extract against all cell line was calculated using the following formula.

The effects of extracts were expressed by IC50 values calculated from dose response curves.
3. Results and Discussion
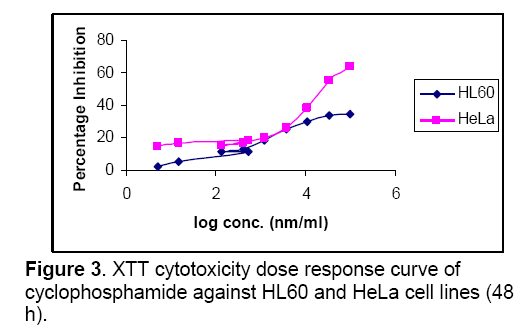
The effect of chloroform,butanol and ethyl acetate extracts of Symplocos racemosa bark (test) and cyclophosphamide (control) on the growth of HeLa and HL60 cell lines were examined by the XTT assay. Dose response curves constructed between the range 0.005 – 100μg/ml and 0.005 - 100μM for S. racemosa extracts and cyclophosphamide (control) respectively,express decreasing number of viable cells with increasing concentration of extract as well as cyclophosphamide. Calculation of IC50 and R2 value was done using graphs generated from Microsoft excel 2003 edition. (Figures 1,2 and 3) The susceptibility of cells to the extract and cyclophosphamide was characterized by IC50 values (Table 1). Results indicate that the cytotoxic effect strengthens with increase in the concentration of extract.
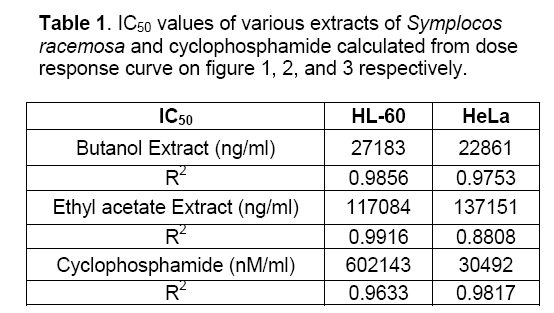
Highest cytotoxicity of butanol extract was found against HeLa cell line,which is more potent than that of cyclophosphamide,which shows that the extract was proven more active against the HeLa than the cyclophosphamide. While in case of ethyl acetate extract highest cytotoxicity was found against HL60 cell line. Butanolic extract also exert the high cytotoxicity against HL60,which is higher than cyclophosphamide cytotoxicity against this cell line. Ethyl acetate extract was also found to be toxic against HeLa cell line. Chloroform extract was found to be non-toxic against both cell lines.
Reason behind such cytotoxicity of butanol and ethyl acetate can be presence of certain phytochemicals such as phenolic glycosides or dithiadiazetidin derivatives. The mechanism behind cytotoxicity can be apoptosis as revealed by structure activity relationship of the phytochemicals present.
4 Conclusions
Different cytotoxicity of Symplocos racemosa extracts against both cancerous cell lines evaluated is based on their chemical composition and relative content of biologically active substance. The literature data are proving the presence of phenolic glycosides,salireposide and benzoyl salireposide,which are found to be present in ethyl acetate extract. The butanol extract has not been explored much to confirm phytochemicals present in it. There are also lots of literature data proving the different pharmacological activity of Symplocos racemosa extract e.g. gonadotropin releasing,antioxidant,antiarthritic and antibacterial [3,8,9].
Further studies are required to determine intracellular pathaway(s) involved in the mechanism of cytotoxicity. This study provides only basic data. Further studies with isolated active substances from extract of analyzed plant should be undertaken.
Acknowledgements
We are thankful to Dr. Ritesh Vaidya,bioscience Department,Ganpat University,Kherva for authentification of the plant.
References
- Madhusudan S.,Middleton M.R. (2005),The immerging role of DNA repair proteins as predictive,prognostic,and therapeutics in cancer.,Cancer Treat. Rev.,31: 603-617.
- Newman D.J.,Cragg G.M.,Snader K.M.,(2003),The natural product as a source of new drugs over the period 1981-2001,Journal of Natural Product.,66: 1022-1037.
- Kumar G.S.,Jayaveera K.N.,Kumar C.S.A. (2007),Antimicrobial effects of Indian medicinal plants against acne-inducing bacteria,Tropical journal of pharmaceutical research ,6: 717-723.
- Abbasi M.A.,Ahmad V.U.,Zubair M. et al. (2004),Phosphodiesterase And Thymidine Phosphorylase-Inhibiting Salirepin Derivatives From Symplocos Racemosa,Planta Med.,,70(12): 1189-94
- Abbasi M.A.,Ahmad V.U.,Zubair M. et al.,(2005),A new alpha.-glucosidase inhibiting dithiadiazetidin derivative from symplocos racemosa,Heterocycles.,65: 1837-1842
- Ahmad V.U.,Zubair M.,Abbasi M.A. et al. (2007),Butyrylcholinesterase Inhibitory C-Glycoside from Symplocos racemosa,Polish Journal of Chemistry ,8:403-407,Bibliogr.
- Freshney R.I.,Culture of animal cells,a manual of basic technique,5th edition,Wiley-Liss,200-1,209-11,213-4,251,328-32,335-8,359-70,508.
- Choudhary M.I.,Fatima N.,Abbasi M.A. (2004),Phenolic glycosides,a new class of human recombinant nucleotidepyrophosphatase phosphodiesterase-1 inhibitors,Bioorganic and medicinal chemistry,12: 5793-5798.
- Bhutani K. K.,Jadhav A. N.,Kalia V. (2004),Effect of Symplocos racemosa Roxb. On gonadotropin release in immature female rats and ovarian histology,Journal of Ethnopharmacology,94(1): 197-200.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences