Cold Pressing Hydrodistillation and Microwave Dry Distillation of Citrus Essential Oil from Algeria: A Comparative Study
Mohamed Amine Ferhat, Mohamed Nadjib Boukhatem, Mohamed Hazzit, Brahim Youcef Meklati, Farid Chemat
Mohamed Amine Ferhat1, Mohamed Nadjib Boukhatem2,3,*, Mohamed Hazzit4, Brahim Youcef Meklati5, Farid Chemat6
1Laboratoire de Recherche sur les Produits Bioactifs et la Valorisation de la Biomasse LPBVB, Ecole Normale Supérieure Vieux Kouba ENS, BP 92, Kouba- Alger, Algeria;
2Département de Biologie et Physiologie Cellulaire, Faculté des Sciences de la Nature et de la Vie, Université Blida 1, Blida, Algeria;
3Laboratoire Ethnobotanique et Substances Naturelles, Ecole Normale Supérieure de Kouba, Alger, Algeria;
4Département de Technologie des Industries Agricoles et Alimentaires, Institut National Agronomique (INA), El- Harrach, Alger, Algeria;
5Centre de Recherches en Analyses Physico-chimiques (CRAPC), Alger RP 16004, Alger, Algeria;
6UMR A 408 INRA, Université d’Avignon, Sécurité et Qualité des Produits d’Origine Végétale, 33 Rue Louis Pasteur, 84029 Avignon cedex 1, France.
Received Date: September 12, 2016; Accepted Date: September 28, 2016; Published Date: October 05, 2016
Citation: Ferhat MA, Boukhatem MN, Hazzit M, et al. Cold Pressing, Hydrodistillation and Microwave Dry Distillation of Citrus Essential Oil from Algeria: A Comparative Study. Electronic J Biol, S:1
Abstract
Hydro-Distillation (HD), Cold Pressing (CP) and Microwave Clevenger or microwave Accelerated Distillation (MAD) methods have been compared and evaluated for their efficiency in the extraction of essential oil (EO) from fresh Citrus peels. EO from peel of 4 varieties of Algerian sweet orange (C. sinensis), Lemons (C. limon), Citrons (C. medica), sour orange (C. aurantium), grapefruits (C. paradisi) and tangelos (C. deliciosa Ten C. tangerina Hort. ex Tan.) × (C. paradisi Macf.) were analysed by Gas Chromatography-Mass Spectrometry (GC-MS). The EO chemical constituents were identified according to their mass spectra and Kováts retention indices determined on polar and non-polar stationary phase capillary columns. Citrus peel oils contained from 86.69 to 99.31% of monoterpenes with limonene reported to be the major component. The Principal Components Analysis (PCA) and the Canonical Discriminant Analysis (CDA) have been undertaken in order to make a comparison between the three processes of extraction (MAD, HD and CP). The MAD offers significant advantages over traditional alternatives, namely; shorter distillation times (30 min. against 3 h for HD and 1 h for CP); improved yields; environmental impact (energy cost is fairly superior to perform HD and for mechanical motor (CP) than that required for rapid MAD extraction); cleaner features (as no residue generation and no water or solvent used) and provides a more valuable EO (with elevated quantities of oxygenated compounds). Hence, it provides also the ability for enhanced reproduction of natural fragrance of the EO from Citrus fruit.
Keywords
Clevenger; Microwave extraction; Essential oil; Hydro-distillation; Green chemistry; Orange peel.
Introduction
The products resulting from the processing of citrus are juices, essential oils and peel. The use of this residue is a fundamental requirement of the fruit processing industry, not only for economical reasons, but also to reduce the severe environmental impact that this could produce [1]. Citrus oils constitute the largest sector of the world production of essential oils (EO). Citrus EOs obtained from the peel of fruit is used in the food and perfume industries. They are mixtures of more than 200 components that can be grouped into 2 fractions which contain monoterpenes and sesquiterpenes hydrocarbons and their oxygenated derivatives along with aliphatic aldehydes, alcohols and esters [2]. Citrus oils are largely employed as aromatizes in the food and pharmaceutical industries. In particular, lemon and bergamot EO are used in the cosmetic industry, for the production of perfumes, detergents and body-care products [1]. Today Citrus fruit, juice or peel oils are currently used in everything from food or food preparations, soft drinks, ice cream, candy, pharmaceutical preparations, air fresheners, cleaning products, solvents as well as colognes and fine perfumes [3].
The quality of Citrus EO obviously depends to large extent on factors (provenance, type of soil, climate, citrus variety), but the processing of the fruit also has a significant effect. EO is complex mixture of volatile substances generally present at low concentrations. Before such substances can be analysed, they have to be extracted from the matrix. The extraction method used has an effect upon the physical properties of Citrus oils. The qualitative characteristics of an EO are almost always closely related to the yield obtained. An exhaustive extraction procedure produces a larger quantity of high-boiling components, with high molecular weights. As a result, the oil has high specific gravity, non-volatile residue and refractive index values, while the optical rotation value is lower because of the lower relative percentage of d-limonene.
In ideal conditions, the extraction of EO would not involve the use of water [4-6]. However, almost all the industrial procedures required the use of water to wash away the oil from the surface of the whole fruit or peel. Since the fragrance of EO is directly related to the content of aldehydes and esters, the amount of water which circulates during processing is very important [1].
The conventional methods for the extraction of Citrus essential oils have some disadvantages. When using cold pressing (CP), Citrus EO is agitated vigorously with water and a gradual diminution in citral and terpene alcohols contents will be observed. Furthermore, during agitation, air is thrashed into the liquid, thereby creating conditions favourable for hydrolysis, oxidation and resinification. For steam distillation and Hydrodistillation (HD), the elevated temperatures and prolonged extraction time can cause chemical modifications of the oil components and often a loss of the most volatile molecules [7,8].
These shortcoming have led to the consideration of the use of new “green” technique in EO extraction, which typically use less solvent and energy, such as supercritical fluids, ultrasound and microwave [9-11]. Microwave energy is a non-ionizing from of electromagnetic radiation that causes molecular motion by migration of ions and rotation of dipoles, but does not normally cause changes in molecular structure. Microwave energy has a frequency range from 300 to 300,000 MHz. The most commonly used frequency for commercial microwave instruments is 2450 MHz, which corresponds to energy output of 600-700 W [12]. The same wavelengths are used for radar and telecommunications transmission.
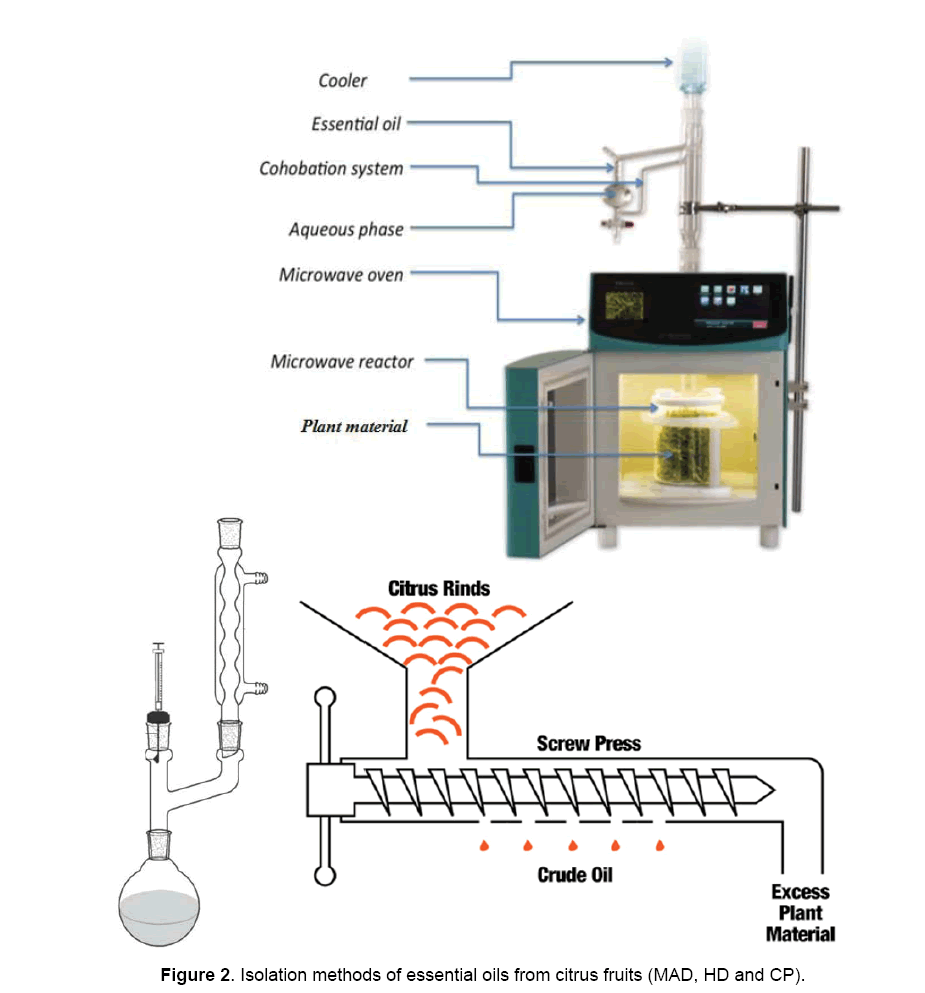
A recent patent describes a new method for extracting natural products without added any solvent or water by using microwave energy [13,14]. Fast microwaveaccelerated distillation or microwave ‘dry’ distillation, known as ‘MAD’ or ‘DryDist’ is an original combination of microwave heating and dry distillation at atmospheric pressure [15,16]. This method involves placing fresh vegetable material in a microwave reactor. The internal heating of the in situ water within the plant material distends it and makes the glands and oleiferous receptacles burst. This process thus frees EO, which is entrained by the in situ water of the plant material by azeotropic distillation. The vapour then passes through a condenser outside the microwave cavity, where it condensed. The distillate is collected continuously in a receiving flask. Excess water is refluxed and recycled to the extraction vessel by cohobation, in order to restore the moisture of the plant material. The EO is collected directly and dried without any added solvent extraction step. MAD has been used to obtain EO from different raw materials.
The aim of this work was to present an improved MAD for the extraction of EO from Citrus peels and compare the results with those obtained by the conventional technique, hydro-distillation (HD) and cold pressing (CP). We have applied MAD, HD and CP techniques to extraction EO from peel of four varieties of Algerian sweet orange (C. sinensis), Lemons (C. limon), Citrons (C. medica), sour orange (C. aurantium), grapefruits (C. paradisi) and tangelos (C. deliciosa Ten C. tangerina Hort. ex Tan.) × (C. paradisi Macf.). We make appropriate comparisons in term of extraction yield and rates, EO composition, and analytical data of chemical compounds were obtained using Principal Components Analysis (PCA) and Canonical Discriminant Analysis (CDA).
Materials and Methods
Plant materials
Table 1 regroups the species and varieties of Citrus studied in this work. Citrus fruits were gathered from the experimental plantation Institut Technique de l’Arboriculture Fruitière (I.T.A.F.), located in the Mitidja region 40 km south of Algiers (Boufarik city, Algeria).
| Species | Varieties | Names of Citrus fruit |
|---|---|---|
| Citrus medica L. | Rhobs-el-arsa | Citrons |
| Citrus limon (L.) Burm. | Eurêka Lemontree Villa França |
Lemons Lemons |
| Citrus sinensis (L.) Osbeck. | Washington Navel Valancia Late Tarocco |
Sweet Oranges Sweet Oranges Sweet Oranges |
| Citrus aurantium | Sorbonne | Sweet Oranges |
| Citrus paradisi | Bouquetier de Nice | Sour Orange |
| (C. deliciosa Ten C. tangerina | Marsh Seedless | Grapefruits |
| Hort. ex Tan) X (C. paradisiMacf.) | Tangelo Seminole | Tangelos |
Table 1. Species and Varieties of Citrus.
Let's note that the variety Washington Navel is part of the navel oranges, Valencia and Sorbonne belong to the white oranges and Taracco is part of blood oranges (Figure 1). Indeed, we chose like basis, the classification of Tanaka that summarized the criteria’s and definition of the names of citrus fruits, in accordance with the more recent international agreements [17].
Citrus fruits were peeled to separate the external part of the lemon (flavedo), giving a yield of 20% (w/w) of the peel with respect to the whole fruit. Fresh plant material was used in all extractions.
MAD apparatus and procedure
MAD was performed using the ‘DryDist’ microwave oven illustrated in Figure 2, (Milestone srl) which is a multimode microwave (MW) reactor (2.45 GHz) with a highest delivered power of 1000 W, variable in 10 W increments. Temperature was controlled using an external Infrared (IR) sensor. In a typical MAD process performed at atmospheric pressure, 200 g fresh lemon peels were heated using a fixed power density of 1 W/g for 30 min without the addition of waters or solvents. A cooling system outside the MW cavity condensed the distillate continuously. Condensed water was refluxed to the distillation vessel to provide identical conditions of temperature and humidity for EO extraction. The distillation was continued at 100°C until no more EO was obtained. The EO was dried over anhydrous sodium sulphate and stored at 4°C until used. Extractions were performed at least three times and the mean values are reported.
Cold pressing apparatus and procedure
EO was collected from 1 kg of lemon fruit, using an automated cold-pressing machine from Schwaub. The epidermis and oil glands were lacerated with a needle, creating areas of compression in the peel surrounded by regions of inferior pressure. The EO was carried down to a decantation vessel in a stream of water, the emulsion being collected and then separated by centrifugation. The EO was collected, dried over anhydrous sodium sulphate and stored at 4°C until used. Extractions were performed at least three times, and the mean values were reported.
Hydrodistillation apparatus and procedure
Fresh lemon peels (200 g) were submitted to HD using a Clevenger-type apparatus (Conseil de l’Europe, 1996) and extracted with 2 l water for 3 h (until no more EO obtained). The EO was collected, dried over anhydrous sodium sulphate and stored at 4°C until used. Distillations were performed at least 3 times and the mean values were reported.
Gas chromatography and gas chromatography– mass spectrometry identification
The EOs were analyzed by gas chromatography coupled to mass spectrometry (GC–MS) (Hewlett– Packard computerized system comprising a 6890 gas chromatograph coupled to a 5973A mass spectrometer) using two fused-silica-capillary columns with different stationary phases. The nonpolar column was HP5MSTM (30 m × 0.25 mm × 0.25 μm film thickness) and the polar one was a StabilwaxTM consisting of CarbowaxTM-PEG (60 m × 0.2 mm × 0.25 μm film thickness). GC–MS spectra were obtained using the following conditions: carrier gas He; flow rate 0.3 mL/min; split-less mode; injection volume 1 μl; injection temperature 250°C; the oven temperature programme was 60°C for 8 min increased at 2°C/min to 25°C and held at 250°C for 15 min; the ionisation mode used was electronic impact at 70 eV. The relative percentage of the components was calculated from GC-FID peak areas. Most constituents were tentatively identified by comparison of their GC Kovats retention indices (RI), determined with reference to an homologous series of C5–C28 n-alkanes and with those of authentic standards available in the authors’ laboratory. Identification was confirmed when possible by comparison of their mass spectral fragmentation patterns with those stored in the MS database (National Institute of Standards and Technology and Wiley libraries) and with mass spectra literature data [18-21].
Statistical analysis
The statistical analyses were carried out using the Microsoft Excel software package (Microsoft Corp.) and XLSTAT add-on (AddinSoft). Principal Components analysis (ACP) and Canonical discriminant analysis (CDA) were performed with extraction methods (MAD, HD and CP) as dependent variable, concentrations of limonene, α-pinene, linalool, (E)-caryophyllene, α-humulene, geranial, valencene, α-terpineol, terpin-4-ol, neral, citronellyl acetate, neryl acetate as explanatory variables.
Results and Discussion
Quantitative analysis
Table 2 represents the yield results in EO of the ten varieties of Citrus fruits obtained by MAD, HD and CP.
| Extraction yield (% g EO/g Citrus fruit) |
|||
|---|---|---|---|
| MAD (30 min) |
HD (180 min) |
CP (90 min) |
|
| Valancia Late | 0.42 | 0.38 | 0.16 |
| Washington Navel | 0.10 | 0.13 | 0.05 |
| Tarocco | 0.17 | 0.15 | 0.04 |
| Sorbonne | 0.29 | 0.35 | 0.01 |
| Villa França | 0.29 | 0.26 | 0.03 |
| Marsh Seedless | 0.10 | 0.11 | 0.03 |
| Rhobs el-arsa | 0.19 | 0.14 | 0.02 |
| TangeloSeminole | 0.22 | 0.22 | 0.04 |
| Bouquetier de Nice | 0.10 | 0.11 | 0.01 |
| Eureka | 0.24 | 0.21 | 0.05 |
Table 2. Essential oils yields of 10 varieties of Citrus fruits obtained by MAD, HD and CP.
In the case of variety Valencia late, Lemon tree Villa Franca and Rhobs el-arsa, the extraction without solvent performed by MAD allows the obtention of EO with yield superior to those obtained by HD, Valencia Late is variety that provides the biggest quantity of EO with the yield of 0.42%. For the rest of the varieties the yield obtained by MAD is similar to those of obtained by HD.
The CP gives only a yield between 0.01 and 0.16%; of way general, it remains appears that for the 10 studied variety; the efficiency in EO exit of the HD is similar to those obtained by MAD, the Valencia Late is the variety that among the 10 studied, offer by MAD of a total length of 30 min, the most important yield.
Composition of the essential oils
The EO of Citrus peels extracted either by MAD, HD or CP are rather similar in their composition and contain the same dominant components (Tables 3 and 4). The terpenes derivatives are present in very big number in all EO of Citrus studied; the limonene is the main derivative. The chemical composition of the EO was the subject of various studies. However, the data of the literature are not always easily comparable because the conditions of extraction are different. The variety" Rhobs el-arsa" coming from the Experimental Station of Boufarik is composed mostly of limonene (93.92-93.19-94.23%) and ß-myrcene (1.63-1.41-1.71%). Of this fact, respectively for an MAD extraction, HD and CP the rate of the other compounds is very weak and lower to 0.65%, especially for nerol (0.47-0.4-0.01%) and geranial (0.30-0.35-0.64%). It is important to note that the linalool, geranial and nerol are only present to very weak contents in the EO obtained by CP.
| N° | Rhobs-el-arsa | Tengelo Seminole | Bouquetier de Nice | Marsh seedless | Sorbonne | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compounds a | MAD | HD | CP | MAD | HD | CP | MAD | HD | CP | MAD | HD | CP | MAD | HD | CP | |
| Monoterpene Hydrocarbons | 96.31 | 95.35 | 96.53 | 97.87 | 98.29 | 99.02 | 94.99 | 96.41 | 99.05 | 93.99 | 95.74 | 97.49 | 96.42 | 98.23 | 98.17 | |
| 1 | α-Thujene | - | - | - | 0.14 | 0.15 | 0.17 | - | - | - | - | - | - | - | - | - |
| 2 | α-Pinene | 0.39 | 0.40 | 0.34 | 0.88 | 0.86 | 0.93 | 0.55 | 0.56 | 0.40 | 0.24 | 0.47 | 0.45 | 0.46 | 0.50 | 0.75 |
| 3 | Sabinene | 0.20 | 0.21 | 0.10 | 0.13 | 0.10 | 0.14 | 0.17 | 0.16 | 0.16 | 0.40 | 0.47 | 0.32 | 0.82 | 0.63 | 0.93 |
| 4 | β-Pinene | - | - | - | 0.47 | 0.38 | 0.37 | 0.59 | 0.62 | 0.61 | - | - | - | - | - | - |
| 5 | β-Myrcene | 1.63 | 1.41 | 1.71 | 1.82 | 1.79 | 1.74 | 1.73 | 1.84 | 1.73 | 1.43 | 1.77 | 1.82 | 1.82 | 1.92 | 1.97 |
| 6 | α-Phellandrene | 0.02 | - | 0.02 | 0.08 | 0.06 | 0.04 | - | - | - | - | - | - | 0.20 | 0.27 | 0.23 |
| 7 | α-Terpinene | - | - | - | 0.06 | 0.07 | 0.08 | 0.02 | 0.01 | - | 0.01 | 0.02 | 0.02 | 0.01 | 0.02 | 0.02 |
| 8 | Limonene | 93.92 | 93.19 | 94.23 | 90.25 | 90.70 | 91.24 | 91.69 | 93.01 | 96.00 | 91.63 | 92.61 | 94.54 | 92.86 | 94.77 | 94.20 |
| 9 | β-(E)Ocimene | 0.15 | 0.14 | 0.13 | 0.20 | 0.20 | 0.18 | 0.24 | 0.21 | 0.15 | 0.27 | 0.35 | 0.34 | 0.05 | 0.05 | 0.07 |
| 10 | γ-Terpinene | - | - | - | 3.65 | 3.78 | 3.95 | - | - | - | 0.01 | 0.05 | - | 0.17 | 0.04 | - |
| 11 | Terpinolene | - | - | - | 0.19 | 0.20 | 0.18 | - | - | - | - | - | - | 0.03 | 0.03 | - |
| Oxygenated Monoterpenes | 1.82 | 1.71 | 1.18 | 1.93 | 1.48 | 0.71 | 1.85 | 2.07 | 0.41 | 0.79 | 0.40 | 0.27 | 1.20 | 1.20 | 0.57 | |
| 12 | Linalool | 0.20 | 0.21 | 0.07 | 0.65 | 0.62 | 0.27 | 1.10 | 1.40 | 0.11 | 0.35 | 0.06 | 0.07 | 0.64 | 0.74 | 0.39 |
| 13 | Citronellal | 0.06 | 0.01 | 0.06 | 0.05 | 0.04 | 0.02 | 0.09 | 0.06 | 0.05 | 0.06 | 0.02 | 0.03 | 0.01 | 0.03 | 0.01 |
| 14 | Terpin-4-ol | 0.03 | 0.04 | - | 0.06 | 0.08 | - | 0.06 | 0.05 | 0.01 | 0.08 | 0.17 | 0.02 | 0.04 | 0.09 | - |
| 15 | α-Terpineol | 0.10 | 0.10 | 0.03 | 0.18 | 0.16 | 0.03 | 0.32 | 0.30 | 0.04 | 0.19 | 0.08 | 0.10 | 0.10 | 0.10 | 0.04 |
| 16 | Nerol | 0.47 | 0.40 | - | - | - | - | 0.07 | 0.08 | 0.01 | - | - | - | 0.05 | 0.03 | - |
| 17 | Neral | 0.21 | 0.20 | 0.38 | 0.23 | 0.09 | 0.09 | 0.10 | 0.17 | 0.16 | 0.04 | - | 0.03 | 0.09 | 0.07 | 0.05 |
| 18 | Geraniol | 0.45 | 0.40 | - | 0.17 | 0.02 | - | 0.10 | - | 0.01 | 0.04 | - | 0.01 | 0.07 | 0.01 | - |
| 19 | Geranial | 0.30 | 0.35 | 0.64 | 0.48 | 0.37 | 0.26 | 0.01 | 0.01 | 0.02 | 0.03 | 0.07 | 0.01 | 0.20 | 0.13 | 0.08 |
| 20 | Thymol | - | - | - | 0.11 | 0.10 | 0.04 | - | - | - | - | - | - | - | - | - |
| Sesquiterpene Hydrocarbons | 0.36 | 0.21 | 0.66 | 0.26 | 0.19 | 0.19 | 0.26 | 0.23 | 0.26 | 0.59 | 0.32 | 0.51 | 0.09 | 0.04 | 0.07 | |
| 21 | (E)Caryophellene | 0.09 | 0.04 | 0.13 | 0.04 | 0.01 | - | 0.10 | 0.14 | 0.11 | 0.23 | 0.20 | 0.28 | - | - | - |
| 22 | α-Humulene | 0.04 | 0.02 | 0.07 | 0.02 | 0.02 | 0.03 | 0.04 | 0.04 | 0.09 | 0.05 | 0.05 | 0.04 | 0.04 | 0.01 | 0.02 |
| 23 | Germacrene D | 0.01 | 0.01 | 0.01 | 0.04 | 0.05 | 0.09 | 0.05 | 0.01 | 0.06 | 0.08 | 0.05 | 0.03 | - | - | - |
| 24 | Valencene | 0.04 | 0.02 | 0.01 | 0.08 | 0.11 | 0.07 | 0.01 | 0.03 | - | 0.23 | 0.02 | 0.16 | 0.03 | 0.01 | 0.03 |
| 25 | β-Bisabolene | 0.18 | 0.12 | 0.44 | 0.08 | - | - | 0.06 | 0.01 | - | - | - | - | - | - | - |
| 26 | Oxygenated Sesquiterpenes | 0.00 | 0.00 | 0.00 | 0.04 | 0.06 | 0.01 | - | - | - | 1.33 | 0.85 | 0.21 | 0.02 | 0.02 | 0.02 |
| 27 | Elemol | - | - | - | 0.03 | 0.05 | - | - | - | - | 0.02 | 0.02 | 0.06 | 0.02 | 0.02 | 0.02 |
| 28 | Caryophellene Alcohol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 29 | Nootkatone | - | - | - | 0.01 | 0.01 | 0.01 | - | - | - | 1.31 | 0.83 | 0.15 | - | - | - |
| 30 | Other Oxygenated Compounds | 0.28 | 0.05 | 0.16 | 0.38 | 0.23 | 0.04 | 1.82 | 0.80 | 0.66 | 0.58 | 0.14 | 0.24 | 0.38 | 0.24 | 0.14 |
| 31 | N-Octanol | - | - | - | 0.10 | 0.16 | - | 0.09 | 0.07 | - | 0.22 | - | - | 0.07 | 0.04 | - |
| 32 | N-Nonanal | 0.02 | 0.04 | - | - | - | - | - | - | - | - | - | - | 0.12 | 0.04 | - |
| 33 | Decanal | 0.03 | - | - | 0.04 | 0.03 | 0.01 | 0.07 | 0.02 | - | 0.34 | 0.13 | 0.16 | 0.14 | 0.14 | 0.13 |
| 34 | Linalyl acetate | - | - | - | 0.08 | - | - | 1.42 | 0.49 | 0.54 | - | - | - | - | - | - |
| 35 | Citronellyl Acetate | 0.02 | 0.01 | - | 0.01 | 0.01 | - | - | - | - | 0.01 | - | 0.04 | 0.02 | 0.01 | 0.01 |
| 36 | Neryl Acetate | 0.09 | - | 0.01 | 0.04 | 0.03 | 0.01 | 0.10 | 0.09 | 0.02 | 0.01 | 0.01 | 0.04 | 0.03 | - | - |
| 37 | Geranyl Acetate | 0.12 | - | 0.15 | 0.11 | - | 0.02 | 0.14 | 0.13 | 0.10 | - | - | - | - | 0.01 | - |
| 38 | Dodecanal | - | - | - | - | - | - | 0.07 | 0.09 | 0.08 | 0.03 | 0.01 | 0.03 | - | - | - |
| Extraction time (min) | 30 | 180 | 120 | 30 | 180 | 120 | 30 | 180 | 120 | 30 | 180 | 120 | 30 | 180 | 120 | |
| Yield % | 0.19 | 0.14 | 0.02 | 0.22 | 0.22 | 0.04 | 0.10 | 0.11 | 0.01 | 0.10 | 0.11 | 0.03 | 0.29 | 0.35 | 0.01 | |
| Total Oxygenated Compounds % | 2.10 | 1.76 | 1.34 | 2.35 | 1.77 | 0.76 | 3.67 | 2.87 | 1.07 | 2.70 | 1.39 | 0.72 | 1.60 | 1.46 | 0.73 | |
| Total non-Oxygenated Compounds % | 96.67 | 95.56 | 97.19 | 98.13 | 98.48 | 99.21 | 95.25 | 96.64 | 99.31 | 94.58 | 96.06 | 98.00 | 96.51 | 98.27 | 98.24 | |
b Retention indices calculated on non-polar HP5MSTM capillary column.
cRetention indices calculated on polar CarbowaxTM-PEG capillary column.
Table 3. Chemical composition of essential oils distillated by MAD, HD and CP extraction from citrus peels (Vrarities of Rhobs-el-arsa, Tengelo Seminole, Bouquetier de Nice, Marsh seedless and Sorbonne).
| N° | Compounds a | Tarocco | Valencia Late | Washington Navel | Eureka | Villa França | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MAD | HD | CP | MAD | HD | CP | MAD | HD | CP | MAD | HD | CP | MAD | HD | CP | ||
| Monoterpene Hydrocarbons | 95.75 | 97.20 | 98.34 | 97.48 | 98.61 | 98.32 | 94.55 | 95.49 | 98.03 | 85.90 | 93.07 | 95.25 | 92.37 | 93.26 | 95.72 | |
| 1 | α-Thujene | - | - | - | - | - | - | 0.01 | 0.08 | - | 0.18 | 0.26 | 0.28 | 0.27 | 0.29 | 0.36 |
| 2 | α-Pinene | 0.43 | 0.47 | 0.50 | 0.43 | 0.53 | 0.51 | 0.42 | 0.53 | 0.50 | 1.00 | 1.34 | 1.31 | 1.15 | 1.37 | 1.56 |
| 3 | Sabinene | 0.63 | 0.51 | 0.45 | 0.54 | 0.49 | 0.54 | 0.75 | 0.34 | 0.54 | - | - | - | - | - | - |
| 4 | β-Pinene | - | - | - | - | - | - | - | - | - | 6.61 | 8.58 | 8.70 | 7.00 | 7.08 | 9.24 |
| 5 | β-Myrcene | 1.69 | 1.65 | 1.84 | 1.64 | 1.87 | 1.82 | 1.65 | 1.76 | 1.87 | 1.09 | 1.57 | 1.62 | 1.53 | 1.52 | 1.69 |
| 6 | α-Phellandrene | 0.19 | 0.13 | 0.22 | 0.15 | 0.17 | 0.36 | 0.19 | 0.12 | 0.15 | 0.02 | 0.05 | 0.03 | 0.05 | 0.03 | 0.05 |
| 7 | α-Terpinene | - | - | - | - | - | - | 0.01 | 0.06 | - | 0.15 | 0.21 | 0.14 | 0.18 | 0.13 | 0.17 |
| 8 | Limonene | 92.71 | 94.39 | 95.32 | 94.64 | 95.48 | 95.06 | 91.39 | 92.49 | 94.89 | 69.65 | 72.90 | 75.68 | 73.99 | 75.78 | 73.75 |
| 9 | β-(E) Ocimene | - | - | - | 0.02 | 0.02 | 0.02 | 0.08 | 0.04 | 0.03 | - | - | - | 0.14 | 0.15 | 0.12 |
| 10 | γ-Terpinene | 0.08 | 0.03 | - | 0.05 | 0.03 | 0.01 | - | - | - | 6.88 | 7.77 | 7.19 | 7.70 | 6.58 | 8.42 |
| 11 | Terpinolene | 0.02 | 0.02 | 0.01 | 0.01 | 0.02 | 0.01 | 0.05 | 0.07 | 0.05 | 0.32 | 0.39 | 0.30 | 0.36 | 0.33 | 0.36 |
| Oxygenated Monoterpenes | 1.59 | 1.07 | 0.77 | 1.09 | 0.55 | 0.55 | 2.79 | 1.25 | 0.79 | 3.33 | 4.37 | 1.96 | 4.96 | 4.43 | 1.84 | |
| 12 | Linalool | 1.02 | 0.68 | 0.44 | 0.62 | 0.27 | 0.30 | 1.86 | 0.42 | 0.15 | 0.19 | 0.30 | 0.10 | 0.30 | 0.32 | 0.12 |
| 13 | Citronellal | 0.03 | 0.02 | 0.02 | 0.06 | 0.03 | 0.04 | 0.07 | 0.04 | 0.05 | 0.03 | 0.05 | 0.03 | 0.11 | 0.05 | 0.07 |
| 14 | Terpin-4-ol | 0.07 | 0.10 | 0.19 | 0.03 | 0.06 | 0.02 | 0.12 | 0.34 | 0.01 | 0.14 | 0.29 | 0.01 | 0.23 | 0.37 | 0.02 |
| 15 | α-Terpineol | 0.14 | 0.10 | 0.04 | 0.10 | 0.06 | 0.05 | 0.22 | 0.26 | 0.03 | 0.37 | 0.39 | 0.15 | 0.43 | 0.53 | 0.14 |
| 16 | Nerol | 0.06 | 0.05 | - | 0.10 | 0.03 | 0.08 | 0.12 | 0.10 | 0.02 | 0.44 | 0.53 | 0.01 | 0.49 | 0.52 | 0.03 |
| 17 | Neral | 0.10 | 0.06 | 0.02 | 0.05 | 0.03 | 0.06 | 0.13 | 0.05 | 0.03 | 0.63 | 0.95 | 0.60 | 1.11 | 0.86 | 0.54 |
| 18 | Geraniol | 0.03 | - | - | 0.04 | 0.01 | - | 0.06 | 0.03 | 0.01 | 0.59 | 0.64 | 0.01 | 0.58 | 0.56 | 0.02 |
| 19 | Geranial | 0.14 | 0.06 | 0.06 | 0.09 | 0.06 | 0.11 | 0.21 | 0.01 | 0.49 | 0.94 | 1.22 | 1.05 | 1.71 | 1.22 | 0.90 |
| 20 | Thymol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| SesquiterpeneHydrocarbons | 0.20 | 0.16 | 0.17 | 0.06 | 0.10 | 0.15 | 0.48 | 0.68 | 0.56 | 0.79 | 0.70 | 0.83 | 0.59 | 0.27 | 0.76 | |
| 21 | E-Caryophellene | 0.03 | 0.01 | 0.02 | 0.02 | 0.02 | 0.01 | 0.04 | 0.03 | 0.03 | 0.27 | 0.21 | 0.23 | 0.18 | 0.16 | 0.21 |
| 22 | α-Humulene | 0.01 | - | 0.01 | 0.01 | 0.03 | 0.03 | - | - | - | - | 0.04 | - | 0.04 | 0.03 | 0.04 |
| 23 | Germacrene D | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 24 | Valencene | 0.14 | 0.14 | 0.12 | 0.02 | 0.05 | 0.02 | 0.08 | 0.38 | 0.30 | 0.06 | 0.08 | 0.02 | 0.04 | 0.05 | 0.01 |
| 25 | β-Bisabolene | - | - | - | - | - | - | 0.04 | 0.02 | 0.01 | 0.44 | 0.37 | 0.58 | 0.33 | 0.30 | 0.50 |
| 26 | Oxygenated Sesquiterpenes | 0.02 | 0.01 | 0.02 | 0.01 | - | 0.09 | 0.32 | 0.25 | 0.22 | 0.02 | - | - | - | - | - |
| 27 | Elemol | 0.02 | 0.01 | 0.02 | 0.01 | - | 0.09 | - | - | - | - | - | - | - | - | - |
| 28 | Caryophellene Alcohol | - | - | - | - | - | - | - | - | - | 0.01 | - | - | - | - | - |
| 29 | Nootkatone | - | - | - | - | - | - | 0.32 | 0.25 | 0.22 | 0.01 | - | - | - | - | - |
| 30 | Other Oxygenated Compounds | 0.36 | 0.26 | 0.26 | 0.50 | 0.27 | 0.31 | 0.94 | 0.36 | 0.33 | 0.97 | 0.84 | 0.67 | 1.10 | 0.78 | 0.71 |
| 31 | N-Octanol | 0.09 | 0.03 | - | 0.15 | 0.05 | - | 0.12 | 0.04 | 0.13 | - | - | - | 0.19 | 0.05 | - |
| 32 | N-Nonanal | 0.05 | 0.01 | - | 0.01 | 0.01 | 0.02 | 0.04 | 0.01 | - | - | - | 0.02 | 0.08 | 0.06 | 0.16 |
| 33 | Decanal | 0.22 | 0.17 | 0.18 | 0.31 | 0.19 | 0.27 | 0.23 | 0.26 | 0.18 | - | 0.07 | 0.01 | 0.02 | 0.02 | 0.02 |
| 34 | Linalyl acetate | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 35 | Citronellyl Acetate | 0.04 | 0.05 | 0.06 | 0.02 | 0.02 | 0.01 | - | - | - | 0.14 | 0.02 | 0.08 | 0.03 | 0.02 | 0.01 |
| 36 | Neryl Acetate | 0.01 | - | 0.02 | 0.01 | - | 0.01 | 0.02 | 0.01 | 0.02 | 0.52 | 0.48 | 0.36 | 0.44 | 0.36 | 0.32 |
| 37 | Geranyl Acetate | - | - | - | - | - | - | 0.53 | 0.04 | - | 0.31 | 0.27 | 0.20 | 0.34 | 0.27 | 0.20 |
| 38 | Dodecanal | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Extraction time (min) | 30 | 180 | 120 | 30 | 180 | 120 | 30 | 180 | 120 | 30 | 180 | 120 | 30 | 180 | 120 | |
| Yield % | 0.17 | 0.15 | 0.04 | 0.42 | 0.39 | 0.16 | 0.10 | 0.13 | 0.05 | 0.24 | 0.21 | 0.05 | 0.29 | 0.26 | 0.03 | |
| Total Oxygenated Compounds % | 1.97 | 1.34 | 1.05 | 1.60 | 0.82 | 0.95 | 4.05 | 1.86 | 1.34 | 4.32 | 5.21 | 2.63 | 6.06 | 5.21 | 2.55 | |
| Total non-Oxygenated Compounds % | 95.90 | 97.36 | 98.51 | 97.54 | 98.71 | 98.47 | 95.03 | 96.17 | 98.59 | 86.69 | 93.77 | 96.08 | 92.96 | 93.53 | 96.48 | |
b Retention indices calculated on non-polar HP5MSTM capillary column.
c Retention indices calculated on polar CarbowaxTM-PEG capillary column.
Table 4. Chemical composition of essential oils obtained by MAD, HD and CP extraction from citrus peels (Varieties of Tarocco, Valencia Late, Washington Navel, Eureka and Villa França).
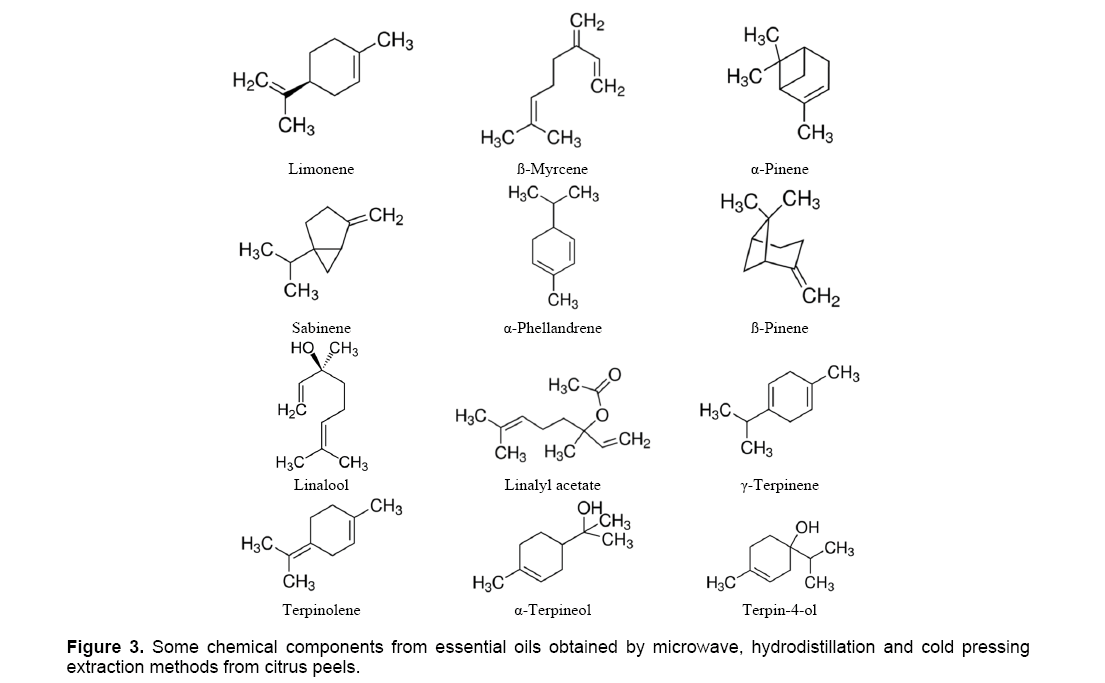
The chemical composition of the EO of the 4 varieties of Citrus sinensis is constituted mainly of compounds hydrocarbon (Figure 3), limonene and ß-myrcene being the most abundant. α-pinene, Sabinene and α-phellandrene are identified in the almost-totality of the varieties. The oxygenated (very minority) fraction, is composed mainly of linalool, of two aldehydes whose contents don't exceed 0.31% (decanal, geranial) and α-terpineol.
The survey achieved on the EO of sour orange zests (Citrus aurantium), variety bouquetier de Nice to hard fruit, be characterized by a majority ultra-content of limonene (91.69-93.01-96%). All composed others necessarily meet respectively with contents weak for an MAD, HD and CP: Among these, ß-myrcene (1.73- 1.84-1.73%), ß-pinene (0.59-0.62-0.61%), α-pinene (0.55-0.56-0.4%) but also of the acyclic oxygenated compounds as the linalool (1.1-1.4-0.11%), linalyl acetate (1.42-0.49-0.54%) and geranyl acetate (0.14-0.13-0.10%).
The chemical composition of the EO of zests of the pomelo is characterized by content in very important limonene (91.63-92.61-94.54%). The hydrocarbon constituent others presents in quantity often substantial are the β-myrcene (1.43-1.77-1.82%), sabinene (0.4-0.47-0.32%), α-pinene (0.24-0.47- 0.45%) and (E)-ß-ocimene (0.27-0.35-0.34%).
One of the main features of citrus fruits is their capacity of hybridization. So many hybrids interspecific or intergeneric exist. Among the more used, the tangelos is descended of the crossing between Citrus deliciosa ten or Citrus tangerine Hort. ex Tan and Citrus paradise Macf. The composition of the EO of the variety Tangelo seminole studied in this work is characterized by very strong contents in limonene (90.25-90.7-91.24%). The other hydrocarbons monoterpenes present in the almost-totality of the samples is on the one hand, the γ-terpinene (3.65- 3.78-3.95%) and ß-myrcene (1.82-1.79-1.74%) identified with substantial contents and on the other hand, α-pinene (0.88-0.86-0.93%), α-thujene (0.14- 0.15-0.17%), sabinene (0.13-0.10-0.14%) and terpinolene (0.19-0.2-0.18%) presents to very weak contents. The oxygenated fraction is composed mainly of linalool (0.65-0.62-0.27%), thymol (0.11- 0.10-0.04%) and one acyclic aldehyde (geranial) whose contents do not pass respectively 0.5% with MAD, HD and CP methods.
We notice a very big likeness in the chemical composition of the EO of zests of the variety" Lemon tree Villa França", and of the one of the Eureka variety. Cyclic monoterpenes constitutes 92.44 to 95.83% of the global composition. After limonene (73.99- 75.78-73.75%), we find about ten compounds whose contents vary from 0.01 to 9.24%: γ-terpinene (7.7- 6.58-8.42%), ß-pinene (7-7.08-9.24%), ß-myrcene (1.53-1.52-1.69%), α-pinene (1.15-1.37-1.56%), terpinolene (0.36-0.33-0.36%),(E)-ß-ocimene (0.14- 0.15-0.12%) and α-thujene (0.27-0.29-0.36%). The oxygenated fraction does not pass 6.81%. To quoted of the geranial (1.71-1.22-0.9%), one finds the neral (1.11-0.86-0.54%), α-terpineol (0.43-0.53-0.14%), terpin-4-ol (0.23-0.37-0.02%), and linalool (0.30- 0.32-0.13).
Classification of the essential oils by Analysis of multi-dimensional data
We used two methods of analysis of multi-dimensional data:
The principal Components Analysis (PCA) and the Canonical Discriminant Analysis (CDA) on the one hand for the statistical survey of the composition of the 30 EO obtained and on the other hand to be able to differentiate the three techniques of extraction MAD, HD and CP [22,23].
PCA is a descriptive method that permits to extract the main components while giving the maximum of information. It describes and classifies the samples studied in spaces of more reduced measurements in order to observe homogeneity or the heterogeneity of the individuals and to know the reason of it. The PCA consists in expressing a set of variables in a set of linear combinations of factors non correlated between them, these factors returning account of a more and more weak fraction of the variability of the data. The representation of the data in spaces of weak dimension, 2 measurements in our case, facilitates the analysis of it considerably.
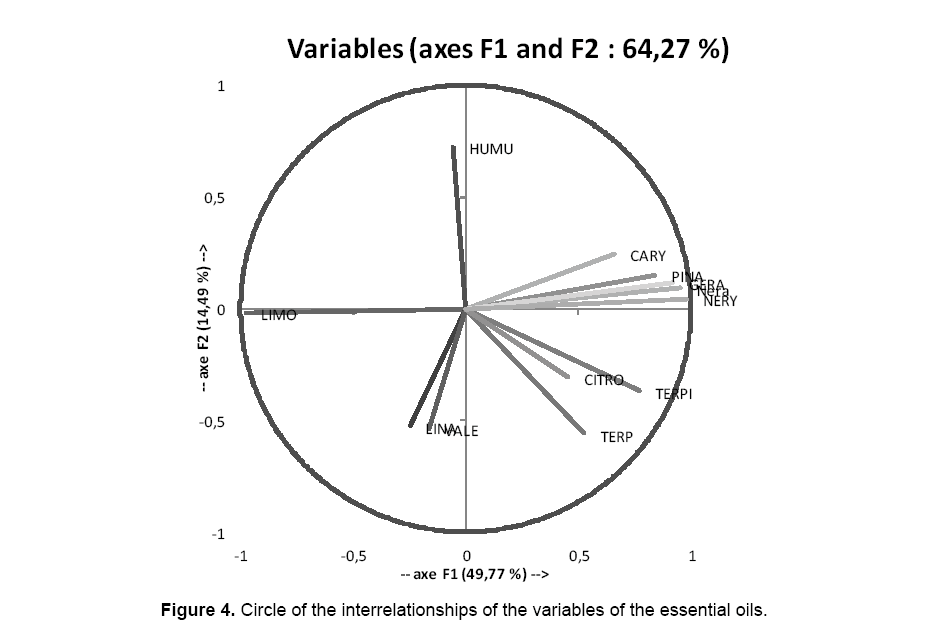
CDA is a method allowing modeling the adherence a group of individuals according to the values taken by several variables, then to determine the likeliest group for an individual, knowing the values of the variables that characterize it solely. The CDA can be considered like an extension of the multiple regression in the case where the variable to explain is a qualitative variable describing some groups. The CDA permits to get a better separation of the groups previously constituted, thanks to discriminative characters that transport the maximum of information. It permits, besides, to affect the individuals to the groups of which they are the nearest [24]. For the treatment of the results, we opted for the software XLSTAT. The chosen type PCA is Pearson, what means that the calculations will be based on a matrix composed of the coefficients of interrelationship of Pearson, coefficients of interrelationship classically used. The first particular diagram to the method is the circle of the interrelationships (to see the circle below on the FI axes and F2). It corresponds to a projection of the initial variables on a plan to two measurements constituted by the first two factors.
When two variables are far from the center of the diagram:
- If they are near one of the other, then they are correlated meaningfully positively (r close to 1).
- If they are orthogonal one in relation to the other, then they are meaningfully non correlated (r close to 0).
- If they are opposed diametrically in relation to the center, then they are correlated meaningfully negatively (r close to -1).
- When the variables are relatively near of the center of the circle of the interrelationship, then all interpretation is hazardous and it is necessary to refer to the matrix of interrelationships to other factorial plans to interpret the results.
In our survey, we could deduct, of the Figure 4 representative the circle of the interrelationships of the variables of the EO, that the FI axes and F2 transport 48.77% and 14.49% of the total information respectively. The circle of the interrelationships is as useful to interpret the significance of the axes. We note that the FI axis is correlated strongly positively with the variable nery (+0.98), nera (+0.95), gera (+0.92), pina (+0.84), terpi (+0.77), cary (+0.66), terp (+0.52), citro (+0.45) and negatively with limo (-0 .97). F2 axis is correlated positively with the variable humu (+0.72), and negatively with vale (-0.53) and lina (-0.52). These tendencies are especially interesting for the interpretation of the diagram of the individuals. To confirm the fact that a variable is bound strongly to a factor, it is sufficient to consult the table of the cosines: more the cosine is raised (in absolute value), more the variable is bound to the axis. More the cosine is close to zero, less the variable is bound to the axis. In our case, we have 3 variables greatly bound in FI nery (0.96), limo (0.95), nera (0.90).
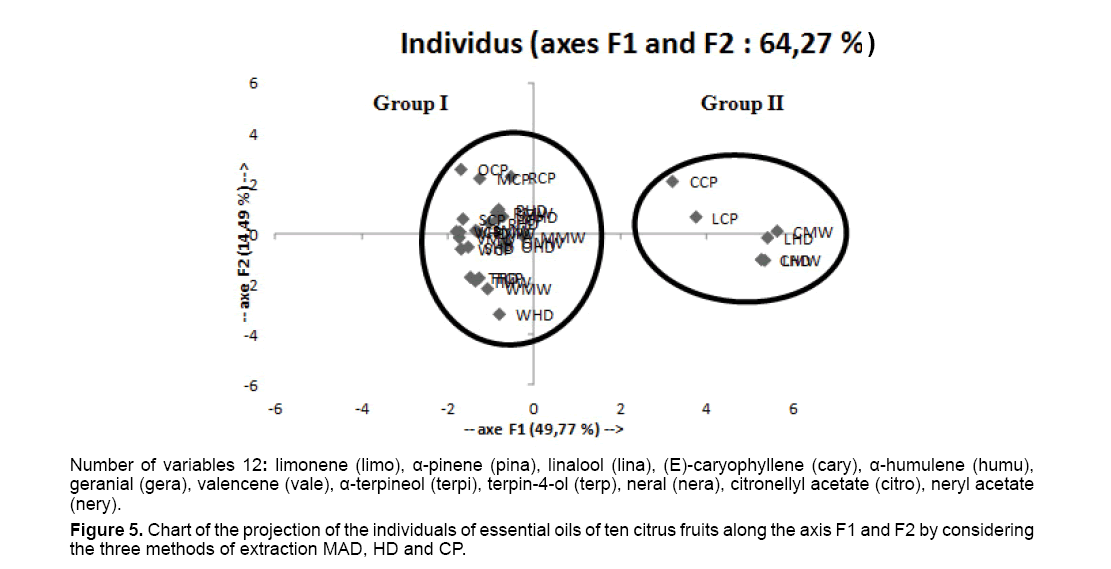
Figure 5 corresponds to one of the objectives of the PCA. It permits to represent the individuals on a plan of projection to two measurements and so to identify the tendencies.
We see in our example that on the basis of the variables of which one arranges, the varieties of studied citrus fruits distribute themselves according to 2 groups according to the FI axis. The I group, is numerically very important which is constituted of 8 varieties (Marsh Seediess, Rhobs el-arsa, Sorbonne, Bouquetier of Nice to hard fruit, Tangelo Seminole, Valancia Late, Washington Navel, Tarocco). The composition of the essential oils is dominated by the limonene (included between 90.25% and 96%). The II group, is only constituted of 2 varieties, the lemon tree Villa França and the Eureka. While comparing these data, we see that the content in limonene doesn't pass 75.78%. On the other hand we observe that other compounds are very discriminative. Thus, the content in α-pinene is superior to 1% in the II group whereas it is lower or equal to 0.9% in the I group. In the same way, the geranial varies from 0.9 to 1.71% in the II group whereas it doesn't pass 0.64% in the I group. We also notice that the rate of neral fluctuates from 0.5 to 1.1% in the II group whereas this compound intervenes with very weak contents (<0.5%) in the composition of all samples of the I group. Finally, the neryl acetate of the II group distinguishes itself of the precedent by a more elevated rate (0.32-0.52%).
The objective of this CDA method is to produce new variables (axes Factorial named also variable discriminative) that separate to best group them from a set of individuals distributed in groups described by several describers. CDA has been achieved on the 10 studied citrus fruits varieties while considering, the relative groups to the methods of extraction in order to put in evidence influence it of every method on the set of the varieties. In our case, we note that two factorial axes have been produced.
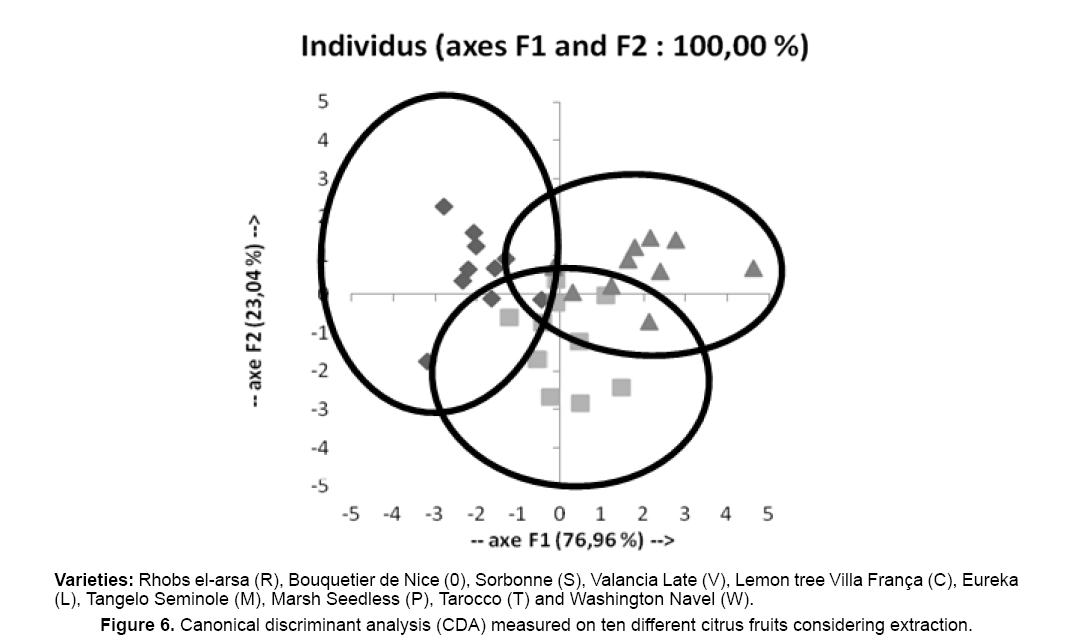
On the Figure 6, the individuals are displayed on the factorial axes. This graph permits to confirm that the individuals are discriminated well on the obtained factorial axes to leave from the initial explanatory variables. It is evident from this survey that the essential oils of zests of 10 varieties of citrus fruits obtained by MAD, HD and CP are characterized so much by a big variability by the nature of the products identified that by their relative proportions.
Canonical Discriminant Analysis (CDA) was performed with extraction methods (MAD, HD and CP) as dependent variable, concentrations of limonene, α-pinene, linalool, (E)-caryophyllene, α-humulene, geranial, valencene, α-terpineol, terpin-4-ol, neral, citronellyl acetate, neryl acetate as explanatory variables. This graph allows us to confirm that extraction procedures are discriminated on factorial axis obtained by using initial explained variables. As a consequence, essential oils extracted from various citrus fruits with MAD, HD and CP methods can be easily discriminated, function of the nature of identified products but also by their relative proportion. Actually, the 30 essential oils studied can be classified in 3 distinct subsections representing MAD, HD and CP procedure respectively. It could be easily seen that the MAD group is relatively closed to CP group and different from the HD group. Indeed, the 30 analyzed essential oils distribute themselves according to 3 distinct groups representing each a technique of extraction among MAD, HD and CP.
Conclusion
The data present in this article demonstrates the potential for Microwave Accelerated Distillation (MAD) in isolation of essential oil from fresh Citrus peels. The beauty of this technique resides in its applicability to a wide variety of samples types.
Microwave Clevenger or Microwave Accelerated Distillation (MAD) makes use of physical and chemical phenomena that are fundamentally different from those applied in conventional solvent extraction and distillation techniques. Using this novel process, fully reproducible extractions can now be completed in minutes without adding solvent or water. Its additional advantages are simplified manipulation and work-up, a higher purity of the final product, no need for a post-treatment of waste water and the consumption of only a fraction of the energy normally required by conventional food extraction methods. All these advantages make MAD a good alternative for the extraction of essential oil from aromatic plants.
References
- Dugo G, Di Giacomo A. (2002). The genuscitrus. Taylor & Francis: London, UK.
- Shaw PE. (1997). Review of quantitative analyses of citrusessential oils. Journal of Agriculture and Food Chemistry. 27:246-257.
- Nonino E. (1997). Where is the citrusindustry going? Parfum and Flavour. 22:53-58.
- Mohapatra HS, Chatterjee A, Kumar P. (2015). Characterization of fibrous assembly from lime peel extract. International Journal of Pharmacology, Phytochemistry and Ethnomedicine. 1:27-36.
- Sandru D, Niculescu V, Lengyel E, et al. (2016). Identification and quantification of total polyphenols in plants with bioactive potentially. International Journal of Pharmacology, Phytochemistry and Ethnomedicine. 4:47-51.
- Ikpa CBC, Ibe FC, Ikpa CU. (2016). Isolation, chemical composition, characterization and anti-bacterial activity of acridinediglycoside from Moringaolifera. International Journal of Pharmacology, Phytochemistry and Ethnomedicine. 2:30-36.
- Pollien P, Fay LB, Chaintreau A. (1998). Simultaneous distillation–extraction: Preparative recovery of volatiles under mild conditions in batch or continuous operations. Flavour and Fragrance Journal. 13:413.
- Luque de Castro MD, Jimenez-Carmona MM, Fernandez-Perez V. (1997). Towards more rational techniques for the isolation of valuable essential oils from plants. Trends in Analytical Chemistry. 18:708-716.
- Reverchon E. Supercritical fluid extraction and fractionation of essential oils and related products. Journalof Supercritical Fluids. 10:1-37.
- Pare JRJ, Belenger JMR. (1997). Instrumental methods in food Analysis, Elsevier, Amsterdam, Netherlands.
- Vinatoru M. (2001). An overview of the ultrasonically assisted extraction of bioactive principles from herbs. Ultrason and Sonochemistry. 8:303–313.
- Kingston HM, Jassie LB. (1998). Introduction to microwave sample preparation. American Chemical Society, Washington, USA.
- Ferhat MA, Meklati BY, Chemat F. (2006). An improved microwave Clevenger apparatus for distillation of essential oils from orange peel. Journal of Chromatography A. 1112:121–126.
- Ferhat MA, Meklati BY, Chemat F. (2007). Comparison of different isolation methods of essential oil from citrus fruits: Cold pressing, hydrodistillation and microwave ‘dry’ distillation,Flavour and Fragrance Journal. 22:494–504.
- Chemat F, Smadja J, Lucchesi ME. (2003). European Patent Application No. EP 1.439.218.A. 2003.
- Chemat F, Smadja J, Lucchesi ME. (2004). US Patent Application No. US 2004.0187340.A1. 2004.
- Tanaka T. (1961). Citologia: Semi centennial commemoration papers on citrus studies, Citologia supporting fondation, Osaka, Japan.
- Arctander S. (1994). Perfume and flavor chemicals. Allured Publishing Corporation, Carol Stream, CA, USA.
- Adams RP. (1995). Identification of essential oil components by gas chromatography mass spectroscopy. Allured Publishing Corporation, Carol Stream, CA, USA.
- Conseil de l’Europe. (1996). Pharmacopée Européenne 1. Maisonneuve S.A. Editions: Sainte Ruffine, France.
- Recueil des Normes Françaises. (2000). Huiles essentielles. Editions AFNOR, Paris, France.
- Mouly PP, Arzouyan CR, Estienne JM. (1995). Chromatographie des flavanonosides des jus de différentes variétés de pamplemousses, différenciation par analyses statistiques multidimensionnelles. Analysis. 23:336-341.
- Mouly PP, Arzouyan CR, Estienne JM. (1996). Différenciation des jus de Citrus par analyses statistiques multivariées. Partie II. Cas des oranges et des mandarines. Analysis. 24:230-239.
- Smadja JM. (1987). Influence climatique et régionale sur la qualité de l’huile essentielle de Vetyver Bourbon. Thèse de Doctorat d’État, Université du Languedoc, France.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences