Salicylic Acid Treatment Extends the Vase Life of Five Commercial Cut Flowers
Hassan Bayat, Mohammad Hossein Aminifard
Department of Horticultural Science, College of Agriculture, University of Birjand, Birjand, Iran.
- Corresponding Author:
- Hassan Bayat
Department of Horticultural Science
College of Agriculture
University of Birjand, Birjand, Iran
Tel: 00989151608215
E-mail: hassanbayat@birjand.ac.ir
Received date: November 30, 2016; Accepted date: January 30, 2017; Published date: February 06, 2017
Citation: Bayat H, Aminifard MH. Salicylic Acid Treatment Extends the Vase Life of Five Commercial Cut Flowers. Electronic J Biol, 13:1
Abstract
To evaluate the efficacy of Salicylic Acid (SA) (0 as control, 100, 200 and 300 mg/l) to extend flower vase life, a completely randomized design with 5 most important cut flowers across the world Alstroemeria peruviana, Gerbera jamesonii, Lilium asiaticum, Rosa hybrida and Polianthes tuberose with 4 replicates and 3 samples for each replicate was conducted. Increasing SA concentrations enhance the vase life to noticeable values and the highest for all cut flowers was observed in 300 mg/L SA treatment. The addition of 300 mg/L SA to clean distilled water extended the vase life of Alstroemeria peruviana, Gerbera jamesonii, Lilium asiaticum, Polianthes tuberose and Rosa hybrida by 40, 67, 21, 71 and 56%, relative to controls, respectively. Salicylic acid treatments increased average petal water content of all cut flowers by 34% in comparison to control. The beneficial effects of salicylic acid are associated with the plant regulating and anti-stress properties of salicylic acid. Results of this experiment provide support for wider use of SA as natural, cheap, safe and biodegradable compound, as a vase solution preservative for prolonging the postharvest longevity of cut flowers.
Keywords
Biodegradable compound; Postharvest; Salicylic acid; Relative fresh weight; Vase life.
Introduction
Cut flowers are valuable products of horticulture. The post-harvest longevity of cut flowers is of critical importance in determining the value of the crop. Many investigations on the vase life and quality of cut flowers have been conducted by adding various preservatives to the vase water causing in cut flower senescence being delayed considerably [1-7]. Floral preservative usually contain germicides, ethylene synthesis inhibitors, growth regulators, some mineral compounds and carbohydrates that are essential to extend the vase life of cut flowers [8].
Flowers maintaining their physiological functions very actively even after harvest and the beginning of their senescence very often depends on ethylene action [9]. There are three limitations which will affect the flower senescence: the water balance, the amount of carbohydrates and the susceptibility to ethylene [7,10-12]. The most important deteriorating factor in cut flower is the blockage of xylem vessels by air and microorganism which caused vascular blockage and thus reduced the vase life of cut flower [13].
Salicylic Acid (SA), a natural plant hormone has important role in abiotic and biotic stress [14]. Salicylic acid is qualified as a plant hormone due to its physiological and biological roles in plants [15]. It has also been suggested as a signal transducer or messenger under stress conditions [16]. Salicylic acid has potential to reduce pH of water and consequently, the proliferation of bacteria was reduced [5,17,18]. It is reported that SA suppresses ACC synthase and ACC oxidase activities and biosynthesis of ethylene in kiwi fruit [19]. Moreover, SA has been shown to restrict with the biosynthesis and/ or action of ethylene in plants [20]. Addition of SA and sucrose to vase solution of cut roses caused a significant reduction in respiration rate, alleviation of the moisture stress and improved the vase life [21]. Moreover, it has been shown that treatment with SA improved postharvest life of different cut flowers [5,10]. Therefore, this experiment was conducted to investigate the effect of SA as components of the vase solution on the vase life of five major economic cut flowers namely Alstroemeria peruviana, Gerbera jamesonii, Lilium asiaticum, Rosa hybrida and Polianthes tuberose.
Materials and Method
Plant material and treatments
Flowering stems of 5 economically important cut flower species namely Alstroemeria peruviana, Gerbera jamesonii, Lilium asiaticum, Rosa hybrida and Polianthes tuberose were supplied from a standard hydroponic greenhouse in Mashhad (36°17’44”N and 59°36’2”E), Iran. Upon arrival in the laboratory, flower stems free of visible damage were trimmed to 45–50 cm-length using cutters. Leaves on the bottom 25 cm of stems that would otherwise be submerged in vase water were removed by hand. Stems were then immediately assigned at random to treatments, depending upon the particular experiment. Glass 800 mL capacity vases were prepared and filled with 300 mL of deionized water and 0, 100, 200 and 300 mg/l SA. Sucrose at 4% was added in all treatments as a base solution.
The opening of each vase was covered in order to limit vase solution evaporative loss and to allow determination of the uptake by stems of the different preservatives and pollution from falling flowers and leaves. Stems were maintained in a controlled environment room at 21 ± 0.5ºC and 60% relative humidity under continuous fluorescent light (47 μmol m−2 s−1).
Vase life determination
Vase life of 5 cut flowers used in this experiment was determined according to the Table 1 [1].
| Species | End of vase life characteristics |
|---|---|
| Alstroemeria peruviana | Wilting and/or drop of >50% of petals |
| Gerbera jamesonii | Drooping of flower and/or moderate wilting of petals |
| Lilium asiaticum | Senescence of all buds on a stem |
| Rosa hybrida | Wilting, bluing, disease of petals and/or drooping of flower |
| Polianthes tuberosa | Wilting and/or petal burning |
Table 1: Criteria used to determine end of flower vase life.
Petal water content
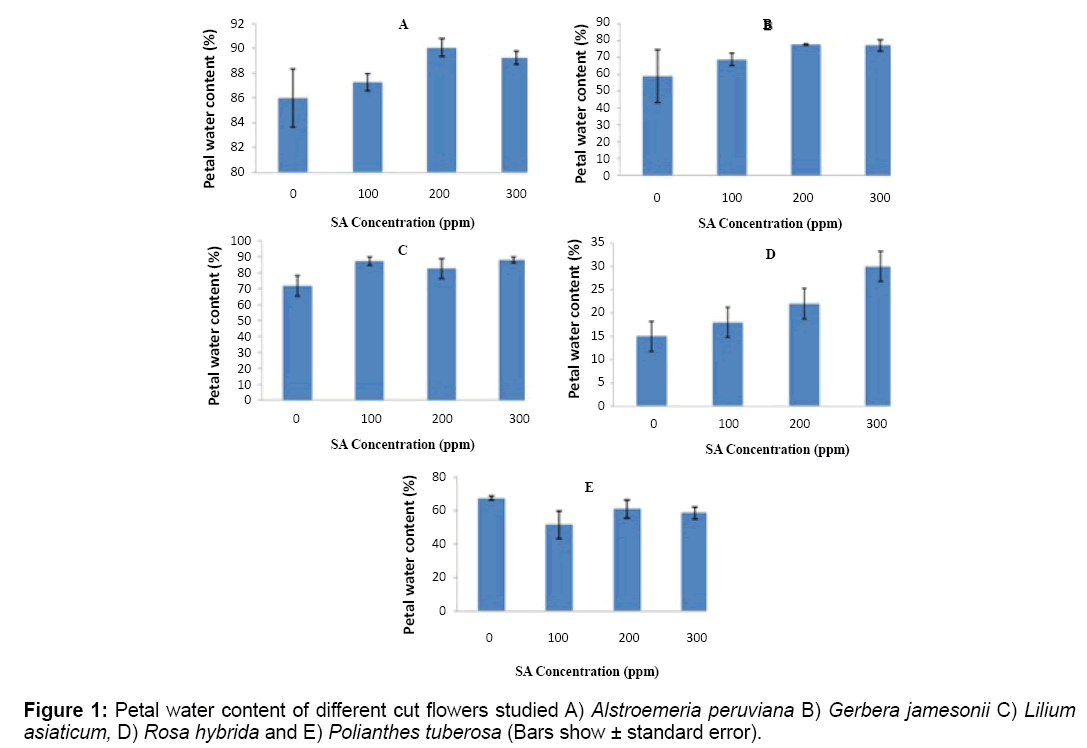
Petal water content from each sample was determined on the end of control treatment vase life for each cut flower studied. For this purpose, 1 g of petals from all replications and each sample were taken as FW and then dried at 70ºC for 24 h and their DW was recorded. Petal Water Content (WP) was then determined with the below equation [22]:
%WP=(FW-DW/DW) × 100
Cut flower water content
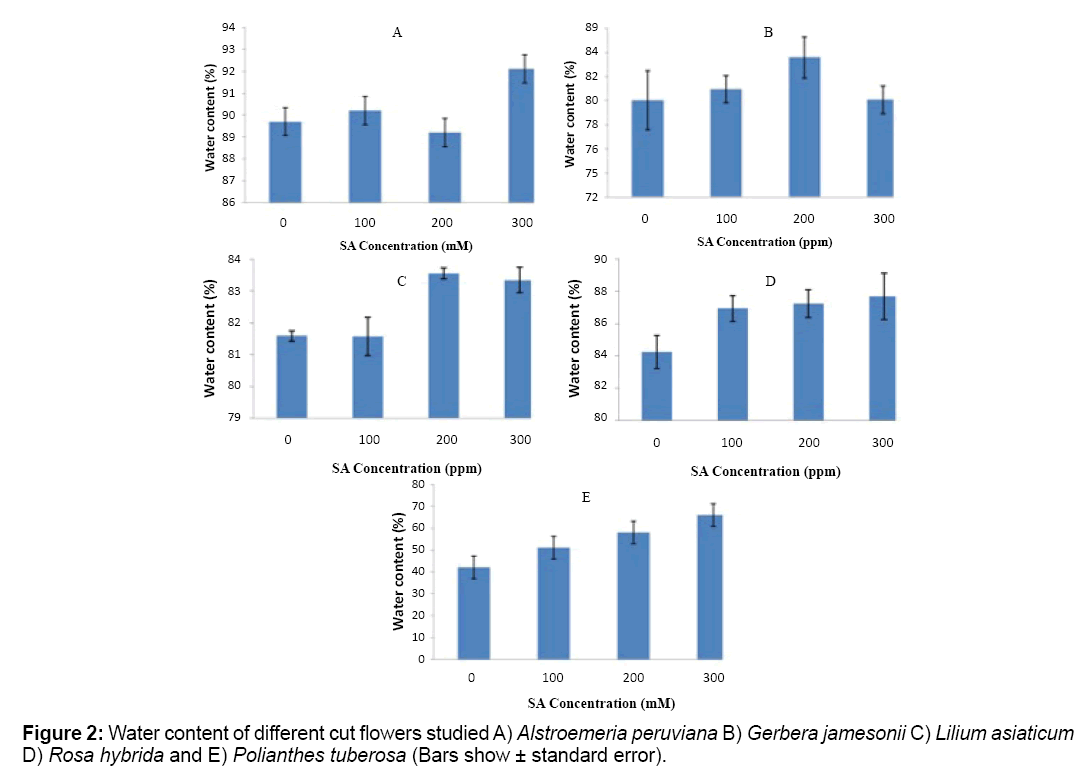
Water contents of the examined cut flowers were similarly determined as petal water contents.
Fresh weight
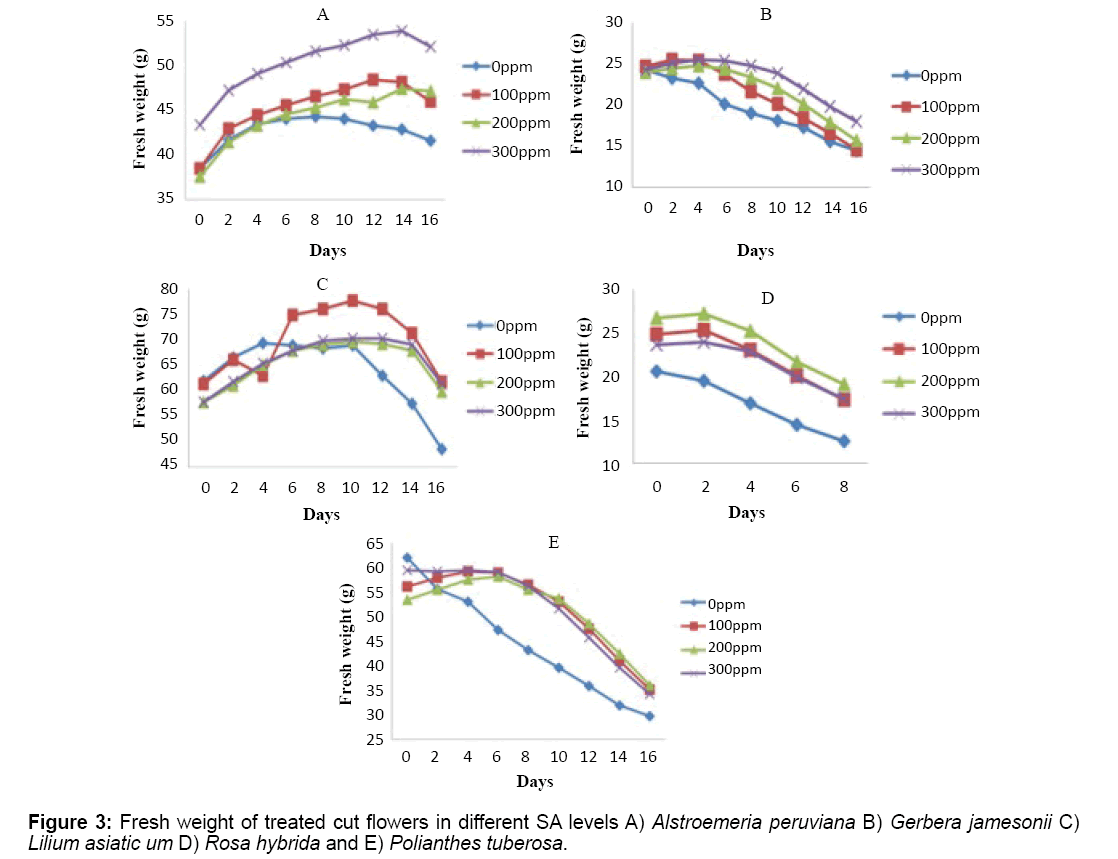
Fresh weight of cut flowers was measured every day through the experiment from day 1 to 9.
Experiment design and data analysis
This experiment was conducted as a completely randomized design with SA at (0, 100, 200, 300 mg/L) with 4 replicates and 3 samples (individual flowers) for each replication. Data were analyzed as completely randomized design ANOVA using JMP8 software. Where significant (P ≤ 0.05) treatment effects were determined by ANOVA, data means were separated by the LSD test.
Results
Vase life
According to the results shown in Table 2, SA as a preservative significantly increased the vase life of all 5 cut flowers (P ≤ 5%). Increasing SA concentrations enhance the vase life to noticeable values and the highest for all was observed in 300 mg/L SA treatment. The addition of 300 mg/L SA to clean distilled water extended the vase life of Alstroemeria peruviana, Gerbera jamesonii, Lilium asiaticum, Polianthes tuberose and Rosa hybrida by 40, 67, 21, 71 and 56%, relative to controls, respectively. Gerbera jamesonii, Rosa hybrid and Polianthes tuberosa showed a greater effect among others. Lilium asiaticum nearly showed the least increasing effect and it seems that senescence goes on with anther release and SA is not able to control this event properly.
| SA concentrations (mg/l) | ||||
|---|---|---|---|---|
| Species | 0 | 100 | 200 | 300 |
| Alstroemeria peruviana | 18.44c* | 22.00bc | 24.11ab | 26.00a |
| Gerbera jamesonii | 10.22c | 11.67bc | 14.00b | 17.11a |
| Lilium asiaticum | 15.55b | 18.00a | 18. 67a | 18.92a |
| Rosa hybrida | 4.96b | 6.11b | 7.14a | 8.51a |
| Polianthes tuberosa | 9.77c | 13.00b | 14.89a | 15.33a |
Table 2: Vase life (day) of tested cut flower species in salicylic acid (SA) treatments.
Petal water content
Results from petal water contents showed significant difference in SA treatments over control (P ≤ 5%). SA treatments with 34% increase by average compared to control, showed the highest petal water content in applied treatments (Figure 1). SA levels were also different significantly for some cut flowers like Rosa hybrida. The lowest amount was measured in control treatment while the highest are differently spread in higher concentrations of (200 or 300 mg/L) within cut flower samples (Figure 1).
Cut flower water content
Placement of cut flowers in vase water containing SA, significantly increased water content at the end of the experiment (P ≤ 1%). The highest amount of this trait was observed in higher concentrations of SA (200 or 300 mg/L) (Figure 2). Alstroemeria peruviana and Lilium asiaticum responded better to the treatments whereas Gerbera jamesonii and Rosa hybrid do not show a regular scheme (Figure 2).
Fresh weight
Effect of SA was highly significant in this trait (P ≤ 1%) and the SA 300 mg/L which had the highest effect among other treatments, even increased the fresh weight in day 14 of the experiment for Alstroemeria peruviana, while a noticeable reduction was observed in control for all cut flowers especially Lilium asiaticum and Polianthes tuberosa (Figure 3). The rate of weight reduction between days 0 to 16 were significantly different in treatments used compared to control. Weight reduction was observed all in cut flowers of this experiment and within higher concentrations as the time to this event goes further on and it happens later. Rosa hybrida lost its weight earlier than others and was only measured to day 8 when the vase life ended (Figure 3).
Discussion
A large number of factors such as pre-harvest conditions, packaging and postharvest handling and storage, interfere with the vase life. Salicylic acid has been found to play a key role in the regulation of plant growth and in the responses to environmental stresses [15,20,23]. Salicylic acid treatments extended vase life in association with inhibition of ethylene production [9,10,24]. Pathogens also affect vase life due to vascular blockage [12]. Salicylic acid has a pH of 2.4 and acidic solution inhibits bacteria growth and proliferation [15,18,25,26]. The addition of SA to vase water has previously been shown to extend the longevity of cut Rosa flowers [18,21].
Water constitutes a large proportion of horticultural products weight. In addition to water, carbohydrates are the other major constituent of these products. These products are commonly take water and other materials from the mother plant, but when cut of, they rapidly move into senescence and death which take place in water loss and weight reduction. This reduction is much higher in stress conditions. Salicylic acid can modulate plant responses to a wide range of oxidative stresses and prevents cell wall degradation [27,28]. As an apparent result in this experiment, in SA treatments after 10 days, no fresh weight reduction is observed and a small increase is even, observed.
Petals of a cut flower are the main ornamental parts and turgidity of this part is important for a good looking product. Petal turgidity depends largely on water uptake and maintenance in treatments used. Results of this experiment showed a significantly higher water uptake and maintain the water in cut flowers which increases the cut flower fresh weight, subsequently. The increases in water uptake and subsequently cut flower fresh weight, is apparently due to the acidifying and stress alleviating properties of SA [5,29]. According to the present results, we can generally discuss that, the major part of the water uptake is gathered in the petals which in fact helps to have a better visual quality in SA treated cut flower samples.
Conclusion
In conclusion, the present study demonstrates that the inclusion of 100-300 mg/l SA in clean vase water can significantly extend the display life of five commercial cut flower species (i.e., Alstroemeria peruviana, Lilium asiaticum, Rosa hybrida, Gerbera jamesonii and Polianthes tuberosa). Higher concentrations showed better effects, although sometimes stem browning was observed at higher concentrations. The present findings provide support for wider testing and use of the natural, cheap, safe and biodegradable compound, SA as a vase solution additive for extending the postharvest longevity of flower species that are susceptible to vascular blockage of bacteria and ethylene.
References
- Abreu ME, Munné-Bosch S. (2008). Salicylic acid may be involved in the regulation of drought-induced leaf senescence in perennials: a case study in field-grown Salvia officinalis L. plants. Environ Exper Bot. 64: 105-112.
- Alaey M, Babalar M, Naderi R, et al. (2011). Effect of pre- and post-harvest salicylic acid treatment on physio-chemical attributes in relation to vase-life of rose cut flowers. Postharvest Biol. Technol. 61: 91–94.
- Alverez AL. (2000). Salicylic acid in machinery of hypersensitive cell death and disease resistance. Plant Mol Biol. 44: 429–442.
- Babalar M, Asghari M, Talaei A, et al. (2007). Effect of pre-and post-harvest salicylic acid treatment on ethylene production, fungal decay and overall quality of Selva strawberry fruit. Food Chem. 105: 449-453.
- Bleeksma HC, van Doorn WG. (2003). Embolism in rose stems as a result of vascular occlusion by bacteria. Postharvest Biol Technol. 29: 334-340.
- Capdeville GD, Maffia LA, Finger FL, et al. (2003). Gray mold severity and vase life of rose buds after pulsing with citric acid, salicylic acid, calcium sulfate, sucrose and silver thiosulfate. Fitopatol Bras. 28: 380-385.
- Elgimabi MN, Ahmed OK. (2009). Effects of bactericides and sucrose-pulsing on vase life of rose cut flowers (Rosa hybirda). Botany Research International. 2: 164-168.
- Gerailoo S, Ghasemnezhad M. (2011). Effect of salicylic acid on antioxidant enzyme activity and petal senescence in ‘Yellow Island’ cut rose flowers. J Fruit Ornam Plant Res. 19: 183-193.
- Gonzalez L, Gonzalez-Vilar M. (2003). Determination of relative water content. In: Handbook of plant ecophysiology techniques. Springer. The Netherlands. 207–212.
- Hayat Q, Hayat S, Irfan M, et al. (2010). Effect of exogenous salicylic acid under changing environment: A review. Environ Exper Bot. 68: 14-25.
- Kang G, Wang C, Sun G, Wang Z. (2003). Salicylic acid changes activities of H2O2-metabolizing enzymes and increases the chilling tolerance of banana seedlings. Environ Exper Bot. 50: 9-15.
- Kazemi M. (2012). Effect of cobalt, silicon, acetylsalicylic acid and sucrose as novel agents to improve vase-life of Argyranthemun flowers. Botany Research International. 2: 164-168.
- Macnish AJ, Leonard RT, Nell TA. (2008). Treatment with chlorine dioxide extends the vase life of selected cut flowers. Postharvest Biol Technol. 50: 197-207.
- Mayak S, Halevy H. (1980). Flower senescence. In: Senescence in plants. Thiman KV. (ed.), CRC, Boca Raton, Florida, USA. 131-156.
- Michael S, Wu MJ. (1992). Ethylene and flower senescence. Plant Growth Regul. 11: 37-43.
- Petridou M, Voyiatzi C, Voyiatzis D. (2001). Methanol, ethanol and other compounds retard leaf senescence and improve the vase life and quality of cut chrysanthemum flowers. Postharvest Biol Technol. 23: 79-83.
- Popova L, Pancheva T, Uzunova A. (1997). Salicylic acid: Properties, biosynthesis and physiological role. Bulg J Plant Physiol. 23: 85-93.
- Raskin I. (1992). Salicylate, a new plant hormone. Plant Physiol. 99: 799-803.
- Redman PB, Dole JM, Maness NO, Anderson JA. (2002). Postharvest handling of nine specialty cut flower species. Sci Hort. 92: 293-303.
- Roein Z, Hassanpour Asil M, Rabiei B. (2009). Silver thiosulphate in relation to vase life of narcissus cut flowers (Narcissus jonquilla). Hortic Environ Biotechnol. 50: 308-312.
- Senaratna T, Touchell D, Bunn E, et al. (2000). Acetyl salicylic acid (Aspirin) and salicylic acid induce multiple stress tolerance in bean and tomato plants. Plant Growth Regul. 30: 157-161.
- Shirasu K, Nakajima H, Rajasekhar VK, et al. (1997). Salicylic acid potentiates an agonist-dependent gain control that amplifies pathogen signals in the activation of defense mechanisms. Plant Cell. 9: 261-270.
- Solgi M, Kafi M, Taghavi TS, et al. (2009). Essential oils and silver nanoparticles (SNP) as novel agents to extend vase-life of gerbera (Gerbera jamesonii cv.‘Dune’) flowers. Postharvest Biol Technol. 53: 155-158.
- Srivastava MK, Dwivedi UN. (2000). Delayed ripening of banana fruit by salicylic acid. Plant Sci. 158: 87-96.
- Vahdati Mashhadian N, Tehranifar A, Bayat H, et al. (2012). Salicylic and citric acid treatments improve the vase life of cut chrysanthemum flowers. J Agr Sci Tech. 14: 879-887.
- van Doorn WG, Zagory D, de Witte Y, et al. (1991). Effects of vase-water bacteria on the senescence of cut carnation flowers. Postharvest Biol Technol. 1: 161-168.
- Zencirkiran M. (2005). Effects of sucrose and silver thiosulphate pulsing on stem-base cracking and vase-life in Leucojum aestivum flowers. J Hortic Sci biotech. 80: 332-334.
- Zencirkiran M. (2010). Effect of 1-MCP (1-Methyl Cyclopropene) and STS (Silver thiosulphate) on the vase life of cut Freesia flowers. Sci Res Essays. 5: 2409-2412.
- Zhang Y, Chen K, Zhang S, et al. (2003). The role of salicylic acid in postharvest ripening of kiwi fruit. Postharvest Biol Technol. 28: 67-74.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences