Neural Stem Cells: A Functional Definition Based on Proteomics
Martin H. Maurer
1Department of Physiology and Pathophysiology,University of Heidelberg,Im Neuenheimer Feld 326,69120 Heidelberg,Germany
2present address: SYGNIS Bioscience GmbH & Co. KG,Im Neuenheimer Feld 515,69120 Heidelberg,Germany
Abstract
Neural stem cells can be isolated from the adult mammalian brain throughout the entire lifetime of an organism, including humans. Unfortunately, no unique molecular marker could be identified expressed by all types of neural stem and progenitor cells in every developmental stage. Genomic and proteomic screening approaches suggest a functional definition of adult stem cells by their molecular inventory. These include (i) the responsiveness to growth factors, (ii) the reuse of developmental signaling cascades in adult progenitors, (iii) the interaction with molecules of the extracellular matrix, (iv) the expression of genes regulating transcription and translation, (v) mechanisms for controlling cell number, and (vi) the protection against cellular stress, such as hypoxia, or DNA damage.
https://sporbahisleri.blogaaja.fi http://sporbahisleri.parsiblog.com https://spor-bahisleri.jimdosite.com https://sporbahisleri.edublogs.org https://sporbahisleri.websites.co.in https://sporbahisleri.podia.com https://sporbahisleri7.wordpress.com https://sporbahisleri.jigsy.com https://niwn-chroiaty-mcieung.yolasite.com https://spor-bahisleri.mywebselfsite.net https://sporbahisleri.mystrikingly.com https://sporbahisleri.splashthat.com https://sporbahisleri1.webnode.com.tr https://sporbahisleri.odoo.com http://sporbahisleri.creatorlink.net http://www.geocities.ws/sporbahisleri/ https://spor-s-site.thinkific.com https://artistecard.com/sporbahisleri https://sporbahisleri.estranky.cz https://spor-bahisleri.mozellosite.com https://651be6b563e56.site123.me https://betsitesiinceleme.blogspot.com https://sporbahisleri.hashnode.dev https://sporbahislerim.wixsite.com/spor-bahisleri https://sporbahislerix.weebly.com https://sites.google.com/view/betsiteleri https://codepen.io/sporbahisleri https://sporbahisleri.bcz.com https://www.smore.com/6rsb9
Keywords
Neural stem and progenitor cell,proteomics,genomics,molecular marker,differentiation.
1. Introduction: Stem and progenitor cells in the brain
In the last 10 to 15 years,the view on the regenerative potential of the brain changed dramatically with the characterization of stem and progenitor cells residing in the adult brain throughout the entire lifetime of the mammalian organism,including humans.
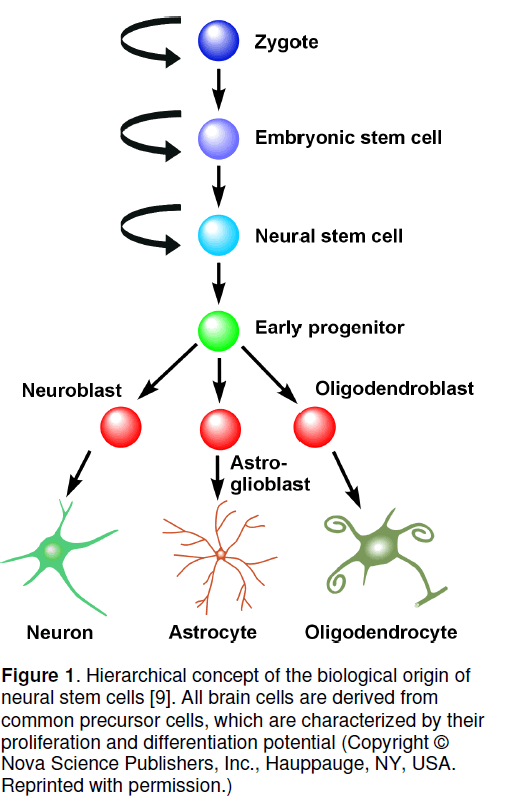
Neural stem and progenitor cells have been characterized by their developmental potential which is the ability to produce progeny with is on the on hand more restricted with regard to the multitude of cells they can produce,but on the other hand gains functionality by differentiation [1-3]. In the current concept,brain cells are all derived from several divisions of (i) the totipotent zygote,which has the potential to give rise to all body cells,via (ii) the pluripotent embryonic stem cells,which cannot constitute the whole body an more,but it can differentiate into all varieties of cells,(iii) the multipotent stem cell,which persists in the organism and gives rise to the cells of the same germinal layer,(iv) progenitor cells of the respective tissue,which are restricted to specific functional cells of the respective organ,and finally,(v) the differentiated tissue-specific cells,which are terminally differentiated and can exert the specific cellular tasks. With regard to the brain,these are neurons,astrocytes,and oligodendrocytes,which are thought to originate all from a common neural progenitor cell (Figure 1) [4-8].
Figure 1. Hierarchical concept of the biological origin of neural stem cells [9]. All brain cells are derived from common precursor cells, which are characterized by their proliferation and differentiation potential (Copyright © Nova Science Publishers, Inc., Hauppauge, NY, USA. Reprinted with permission.)
2. The search for common molecular markers of neural stem cells
Whereas hematopoietic stem cells (HSCs) can be defined by the expression of a specific set of molecular markers,such as the typical combination of CD34+,CD59+,Thy1/CD90+,CD38lo/-,Ckit/ CD117-/lo,lin- for human hematopoietic stem cells [10-12]. However,also HSCs may be present in the bone marrow without this typical phenotype.
Unfortunately,no such definition has been found for neural stem cells [6,13,14],thus the definition of these cells relays on the functional properties self-renewal and multipotency [5,15]. Although presumptive markers have been proposed (reviewed in [16]),none of them comprehends all subtypes of neural stem cells at any given point of time or region in development (Figure 2 ).
Figure 2. Pseudoclustering of molecular markers expressed in neural stem cells and their progeny. There are no unique proteins expressed, therefore, a definition solely based on these markers is not possible. (blue boxes: expression confirmed, light-blue boxes: expression disputed, red box: expression unclear) (GRP, glialrestricted precursors) (Modified from [9]).
In microarray-based screening approaches,specific gene expression patterns have been proposed for the differentiation stages of neural stem cells [17,18]. Genomic approaches also defined the overlapping set of expressed genes in several types of stem cells,including neural stem cells [19-22].
3. Definiton of adult stem cells by common functional properties
With regard to proteomic analyses,proteomic inventories of neural stem cells have been created using two-dimensional gel electrophoresis [23,24]. Additionally,the events underlying cellular differentiation in vitro have been studied in human fetal cortex [25],rodent subventricular zone [26-28],hippocampus [23,29],olfactory bulb [30],and cerebellum [31,32],as well as in neural stem celllike cell lines [33,34] and the porcine brain [35].
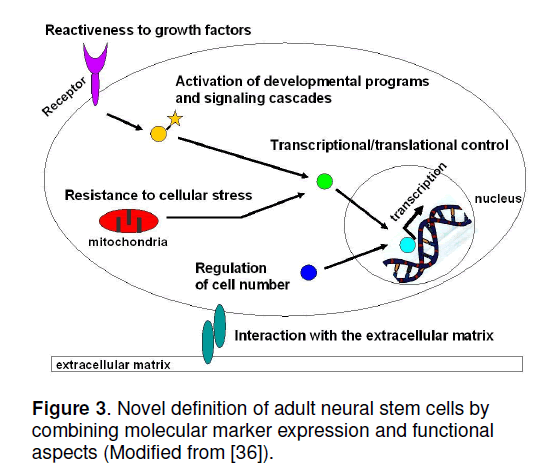
Comparing the protein expression patterns,no single characteristic molecule could be identified,but the functional definition,as specified for the genomic approaches,is supported (Figure 3 ) [9,36]. In the light of these results,adult stem cells have all the same prerequisites,but the exact molecular composition dependent on cell and tissue-specific factors. These common prerequisites involve:
(i) The responsiveness to growth factors such as EGF,FGF-2,EPO,G-CSF,VEGF,LIF,TGF-ß,NGF,as well as to neurotransmitters such as glutamate,GABA,or NO. Again,not all neural stem and progenitor cells react to each of these molecules,but,for example,the expression of VEGF receptors in the rodent brain is highest in the zones of spontaneous neurogenesis,i. e. hippocampus,subventricular zone,and olfactory bulb [37]. A prerequisite to growth factor responsiveness is the expression of specific membrane-spanning receptors,which are able to initiate intracellular signaling cascades.
(ii) The reuse of developmental signaling cascades in adult progenitors. Nearly 20 such pathways have been identified in neural stem cells,including Shh,Pax,Hox,Wnt,Notch/Delta,TGF-ß,NFkappaB,or JAK/STAT [38]. Interestingly,the activation of these pathways in the adult may have different functions compared to the embryonic developmental pathways.
(iii) The interaction with molecules of the extracellular matrix. During differentiation and maturation,stem cells migrate,extend processes,attach to the extracellular matrix,and adhere to nearby microenvironmental surfaces. Therefore,they require the expression of motor proteins,lytic enzymes,and enzymes for providing metabolic energy. Several extracellular matrix proteins mediating cell-cell interaction have been identified in neural stem cells,such as integrins and cadherins.
(iv) The expression of genes regulating transcription and translation. Although no specific proteins in the categories of transcription factors,DNA or RNA binding proteins,and chromatin remodeling enzymes can be named,stem cells need these molecules to change the cellular phenotype from its undifferentiated form to the new functional requirements of a differentiated stem cell.
(v) Mechanisms for controlling cell number. Mainly three processes regulate stem numbers: Mitosis (proliferation),differentiation,and apoptosis (programmed cell death). An essential process is also asymmetric cell division,which regulates the stem cell pool. In this context,typical proteins regulating cell number are caspases and cyclins.
(vi) The protection against cellular stress,which involves metabolic deprivation,NO and O2 toxicity,DNA damage,mechanical distortion,hypo- and hyperthermia,or hypoxia. Proteins in this category comprehend molecular chaperones,NO detoxifying enzymes,and components of the proteasome.
Of note,although adult neural stem cells express these molecules,different proteins can be found in the cells. This hetereogeneity in the protein expression pattern is supported by protein expression studies comparing neural stem and progenitor cells from different brain regions [30,39,40].
4. Conclusions
Although it is not possible to identify a single molecular marker expressed by all types of neural stem cells at any given time in development and adulthood,nor a specific set of expressed markers fulfilling these requirements,it is possible to define neural stem and progenitor cells by common functional properties. A major advance in the field was the introduction of functional genomic / proteomic approaches,mainly based on twodimensional gel electrophoresis for protein separation and mass spectrometry for protein identification.
References
- Weissman I.L. (2000) Stem cells: units of development,units of regeneration,and units in evolution. Cell,100(1): 157-168.
- Hall P.A.,Watt F.M. (1989) Stem cells: the generation and maintenance of cellular diversity. Development,106(4): 619-633.
- Potten C.S.,Loeffler M. (1990) Stem cells: attributes,cycles,spirals,pitfalls and uncertainties. Lessons for and from the crypt. Development,110(4): 1001-1020.
- Gage F.H. (2002) Neurogenesis in the adult brain. J Neurosci,22(3): 612-613.
- Gage F.H. (2000) Mammalian neural stem cells. Science,287(5457): 1433-1438.
- Rakic P. (2002) Adult neurogenesis in mammals: an identity crisis. J Neurosci,22(3): 614-618.
- Galli R.,Gritti A.,Bonfanti L.,et al. (2003) Neural stem cells: an overview. Circ Res,92(6): 598-608.
- Temple S.,Qian X. (1996) Vertebrate neural progenitor cells: subtypes and regulation. Curr Opin Neurobiol,6(1): 11-17.
- Maurer M.H.,Kuschinsky W. (2006) Towards a molecular signature of neural stem cells. In Neural Stem Cell Research (Greer E.V.),Nova Science Publishers,Hauppauge,NY,51-76.
- Bonnet D. (2002) Haematopoietic stem cells. J Pathol,197(4): 430-440.
- Jackson K.A.,Majka S.M.,Wulf G.G.,et al. (2002) Stem cells: a minireview. J Cell Biochem Suppl,38(1- 6.
- Weissman I.L.,Anderson D.J.,Gage F. (2001) Stem and progenitor cells: origins,phenotypes,lineage commitments,and transdifferentiations. Annu Rev Cell Dev Biol,17(387-403.
- Kornblum H.I.,Geschwind D.H. (2001) Molecular markers in CNS stem cell research: hitting a moving target. Nat Rev Neurosci,2(11): 843-846.
- Eiges R.,Benvenisty N. (2002) A molecular view on pluripotent stem cells. FEBS Lett,529(1): 135-141.
- Imitola J.,Snyder E.Y.,Khoury S.J. (2003) Genetic programs and responses of neural stem/progenitor cells during demyelination: potential insights into repair mechanisms in multiple sclerosis. Physiol Genomics,14(3): 171-197.
- Bazan E.,Alonso F.J.,Redondo C.,et al. (2004) In vitro and in vivo characterization of neural stem cells. Histol Histopathol,19(4): 1261-1275.
- Wen X.,Fuhrman S.,Michaels G.S.,et al. (1998) Large-scale temporal gene expression mapping of central nervous system development. Proc Natl Acad Sci USA,95(1): 334-339.
- Gurok U.,Steinhoff C.,Lipkowitz B.,et al. (2004) Gene expression changes in the course of neural progenitor cell differentiation. J Neurosci,24(26): 5982-6002.
- Ivanova N.B.,Dimos J.T.,Schaniel C.,et al. (2002) A stem cell molecular signature. Science,298(5593): 601-604.
- Ramalho-Santos M.,Yoon S.,Matsuzaki Y.,et al. (2002) "Stemness": transcriptional profiling of embryonic and adult stem cells. Science,298(5593): 597-600.
- Burns C.E.,Zon L.I. (2002) Portrait of a stem cell. Dev Cell,3(5): 612-613.
- Li L.,Akashi K. (2003) Unraveling the molecular components and genetic blueprints of stem cells. Biotechniques,35(6): 1233-1239.
- Maurer M.H.,Feldmann R.E.,Jr.,Fütterer C.D.,et al. (2003) The proteome of neural stem cells from adult rat hippocampus. Proteome Sci,1(1): 4.
- Unwin R.D.,Gaskell S.J.,Evans C.A.,et al. (2003) The potential for proteomic definition of stem cell populations. Exp Hematol,31(12): 1147-1159.
- Pearce A.,Svendsen C.N. (1999) Characterisation of stem cell expression using two-dimensional electrophoresis. Electrophoresis,20(4-5): 969-970.
- Salim K.,Guest P.C.,Skynner H.A.,et al. (2007) Identification of proteomic changes during differentiation of adult mouse subventricular zone progenitor cells. Stem Cells Dev,16(1): 143-165.
- Salim K.,Kehoe L.,Minkoff M.S.,et al. (2006) Identification of differentiating neural progenitor cell markers using shotgun isobaric tagging mass spectrometry. Stem Cells Dev,15(3): 461-470.
- Maurer M.H.,Brömme J.O.,Feldmann R.E.,Jr.,et al. (2007) Glycogen Synthase Kinase 3beta (GSK3beta) Regulates Differentiation and Proliferation in Neural Stem Cells from the Rat Subventricular Zone. J Proteome Res,6(3): 1198-1208.
- Maurer M.H.,Feldmann R.E.,Fütterer C.D.,et al. (2004) Comprehensive proteome expression profiling of undifferentiated vs. differentiated neural stem cells from adult rat hippocampus. Neurochem Res,29(6): 1129-1144.
- Murer M.H.,Feldmnn R.E.,Jr.,Burgers H.F.,et l. (2008) Protein expression differs between neurl progenitor cells from the dult rt brin subventriculr zone nd olfctory bulb. BMC Neurosci,9(1): 7.
- Chtterjee M.,Chtterjee D. (2005) Developmentl chnges in the neuronl protein composition: study by high resolution 2D-gel electrophoresis. Mol Cell Biochem,272(1-2): 201-207.
- Tok M.,Wkmiy .,Nkym H.,et l. (2000) Protein profiling of rt cerebell during development. Electrophoresis,21(9): 1872-1879.
- Hoffrogge R.,Beyer S.,Hubner R.,et l. (2007) 2-DE profiling of GDNF overexpression-relted proteome chnges in differentiting ST14 rt progenitor cells. Proteomics,7(1): 33-46.
- Hoffrogge R.,Mikkt S.,Schrf C.,et l. (2006) 2-DE proteome nlysis of proliferting nd differentiting humn neuronl stem cell line (ReNcell VM). Proteomics,6(6): 1833-1847.
- Sklnikov H.,Hld P.,Vodick P.,et l. (2007) proteomic pproch to studying the differentition of neurl stem cells. Proteomics,7(11): 1825-1838.
- Murer M.H.,Kuschinsky W. (2006) Screening the brin: moleculr fingerprints of neurl stem cells. Curr Stem Cell Res Ther,1(1): 65-77.
- Murer M.H.,Tripps W.K.C.,Feldmnn R.E.,et l. (2003) Expression of Vsculr Endothelil Growth Fctor nd its receptors in rt neurl stem cells. Neurosci Lett,344(3): 165-168.
- Gerhrt J. (1999) 1998 Wrkny lecture: signling pthwys in development. Tertology,60(4): 226-239.
- Ostenfeld T.,Joly E.,Ti Y.T.,et l. (2002) Regionl specifiction of rodent nd humn neurospheres. Brin Res Dev Brin Res,134(1-2): 43-55.
- Hitoshi S.,Tropepe V.,Ekker M.,et l. (2002) Neurl stem cell lineges re regionlly specified,but not committed,within distinct comprtments of the developing brin. Development,129(1): 233-244.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences