Molecular Cytogenetics of some Anopheles Mosquitoes (Culicidae: Diptera)
Monika Sharma, S. Chaudhry
Mosquito Cytogenetics Unit, Department of Zoology, Panjab University,Chandigarh 160 014, India.
Abstract
Experimental work pertaining to the molecular cytogenetics of malaria vector species of mosquitoes by the application of PCR technique has been carried out. The main objectives of the study included the sequence characterization of nuclear rDNA internal transcribed spacers 1 and 2 (ITS1, ITS2) and mitochondrial DNA COII gene as potential molecular markers for studying genetic relatedness and phylogenetic kinship among six important species of genus Anopheles. The present studies involved the extraction of genomic DNA from a single female mosquito followed by its amplification with specific primers. The total length of each DNA band with respect to the number of nucleotides was calculated along with GC:AT content, ratio of substitutions due to transitions and transversions (ts/tv), insertions/ deletions and identification of tandem and nontandem repeat sequences. Out of the three studied molecular markers, ITS1 and ITS2 were GC rich while COII gene was AT rich. As for the incidence of insertions/deletions (indels) of bases is concerned, it was found maximum in ITS1 and ITS2 and minimum in conserved COII gene sequence .From the present results it was evident that except for An. culicifacies the ITS2 sequence of the remaining five species is under rapid evolutionary changes due to less conserved nature of the sequence. To the contrary ITS1 sequence was found to have highly variable length ranging from 300-900bp. It was found that An. stephensi + An. culicifacies and An. annularis + An. splendidus share a close genetic homology while An. subpictus and An. maculatus have hypervariable non-homologous genomic qualities. The phylogenetic dendrograms revealed that An. stephensi and An. culicifacies grouped together only when analysed by MP in case of ITS1 gene only while they drifted apart in case of ITS2 and COII gene. This is attributed to their host and habitat feeding preferences which have given them the dubious status of urban and rural vectors respectively.
Keywords
Phylogeny; Anopheles; ITS1; ITS2; COII.
1. Introduction
In integrated mosquito control programmes, taxonomic and phylogenetic studies had been quite useful in understanding the genetics of vectorial capacity and insecticide resistance. In the recent years the developments in DNA based molecular cytogenetics have offered promising possibilities for extension and future applications of genetic engineering based vector control programmes. Some of the molecular level genomic studies involving in vitro amplification of DNA by using the technique of polymerase chain reaction (PCR) have revealed that eukaryotic organisms have considerable nuclear and mitochondrial DNA polymorphism which provides virtually unlimited opportunities for establishing the exact taxonomic status and phylogenetics of species [1-4].
One of the most widely used regions of the genome to infer genetic variations and phylogenetic relationships is the rDNA gene cluster of tandemly repeated multigene family. This multigene family is known to evolve cohesively within species through concerted evolution, a mechanism that tends to homologize sequences within species with simultaneous variations amongst them [5-8]. In between the gene sequences coding for 18S, 5.8S and 28S rRNA are the non-coding internal transcribed spacers 1 and 2 whose sequences are being used for detecting micro and macro geographic genomic variations between species.
In addition to the rDNA domain, the mitochondrial DNA is also being exploited for comparative genomics. In fact, analysis of the parts of mt. DNA and direct sequencing of its specific regions are currently the methods of choice for majority of the population level studies [9-11]. The small size of mitochondrial genome, its single copy number, lack of introns and maternal inheritance, are some of its features for which it is preferred for DNA diagnostics.
2. Materials and Methods
Motivated by the advances made in molecular cytogenetics of mosquitoes, the present topic of research on molecular cytogenetics of some Anopheles mosquitoes (Diptera: Culicidae) was undertaken to carry out the sequence based phylogenetic inferences of six epidemiologically important species of subgenus Cellia of genus Anopheles viz: An. stephensi, An. culicifacies, An. maculatus, An. subpictus, An. annularis and An. splendidus. Out of them, An. stephensi, An. splendidus, An. maculatus and An. annularis belong to Neocellia series, An. subpictus belongs to Pyretophorus series while An. culicifacies belongs to Myzomyia series.
The main objective of the study was the sequence characterization of ITS1, ITS2 and COII gene as potential molecular markers. Cx. quinquefasciatus was used as an outgroup, so as to validate the results. The field collection of these species was carried out from villages Beladhayani near the township of Nangal, Punjab (105 kms North-west of Chandigarh) and Nadasahib, Panchkula (Haryana), 20 kms South-east of Chandigarh (30º44"N, 76º53"E). All these species are vectors of malaria in different capacities, out of which An. stephensi and An. culicifacies are rated as chief urban and rural vectors respectively. The identification of each species was carried out from their morphotaxonomic characters and the species-specific banding pattern of the salivary polytene X-chromosome. Each gravid female was handled in a test tube where it was allowed to lay eggs on a strip of wet filter paper.
In order to provide the optimal conditions of rearing, the eggs were transferred to various bowls and kept in BOD incubator. The freshly emerged unfed adults were stored at –200C before using them for DNA extraction. The extraction of DNA from a single female mosquito at a time was carried out by phenol-chloroform extraction method. The PCR was programmed for 35 cycles of denaturation, annealing and extension by using specific forward and reverse primers meant for ITS1, 2 and COII viz: ITS1- FP- 5’-CCTTTGTACACACCGCCCGT-3’, RP- 5’- GTTCATGTGTCCTGCAGTTCAC - 3’; ITS2- FP - 5’ – TGTGAACTGCAGGACACAT - 3’, RP - 5’ – TATGCTTAAATTCAGGGGGT - 3’; COII – FP - 5’ – TCTAATATGGCAGATTAGTGC - 3’, RP: 5’ – GATCATTACTTGCTTTCAG - 3’. The amplified products were then subjected to 2% agarose gel electrophoresis along with a standard DNA ladder (Gene ruler) of 80-1031bp. The DNA of each band generated from ITS1, ITS2 and COII were sequenced and the sequence alignment data was recorded for total bp composition, GC: AT content, ratio of substitutions (ts/tv) content, insertions/ deletions (indels), incidence of dimers, trimers, tetramers and polymers, and the presence of tandem and non-tandem repeats.
Phylogenetic relationships among the six species and the dendrograms of genetic relatedness were generated by the application of maximum parsimony (MP) method which is based on the assumption that mutation is rare and the best explanation of evolutionary history is the one that requires the least mutation and Kimura-2 parameter.
3. Results
3.1 Sequence analysis of ITS1
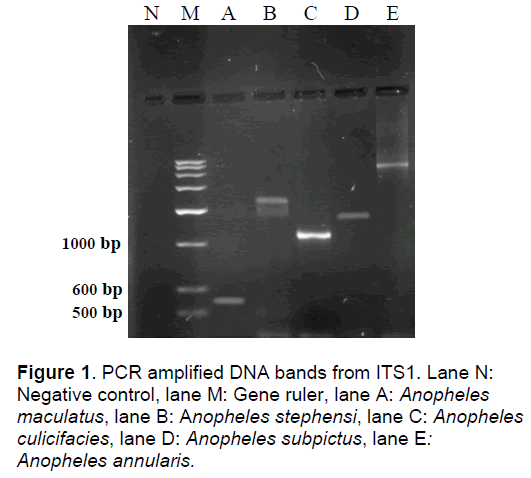
The ITS1 region lying between 18S and 5.8S rRNA coding sequences in the rDNA domain of nuclear DNA yielded G:C rich DNA fragments ranging in length from 300-900 bp as they consisted of 639, 512, 346, 778 and 816 bp in An. stephensi, An. culicifacies, An. maculatus, An. subpictus and An. annularis respectively (Figure 1).
There were a total of 393 substitutions, out of which 174 were transitions while 199 were transversions. The sequence alignment analysis revealed that ts/tv ratio ranged from 0.55 to 1.2 with 0.55 in An. maculatus, 0.83 in An. culicifacies, 0.91 in An. annularis, 0.93 in An. subpictus and 1.2 in An. stephensi. Indels were observed at several places along the sequence in which minimum incidence of loss or gain of base was found in An. annularis (16) and maximum in An. maculatus (51) out of the total of 191 places where insertions or deletions of bases had taken place. So far, the sequence analysis of ITS1 has been carried out only in a few species of genus Anopheles where considerable variation has been found in its length. For example, the ITS1 of An. aconitus has an average length of 503 bp while An. farauti has as many as 979 bp. Outside the genus Anopheles, in Aedes aegypti its length was found to be 418 bp while in the related dipteran Drosophila arizonae this region varied from 500-600 bp. When all the species were compared, it was found to be longest in An. farauti and smallest in An. maculatus, a condition that demands the study of intron based evolutionary patterns in the genus.
3.2 ITS1 sequence based phylogenetics
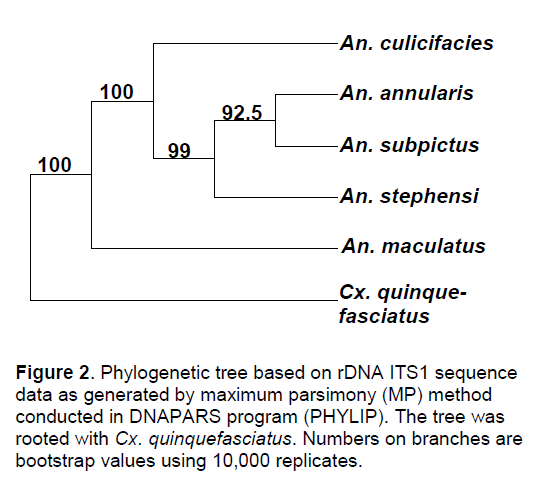
With the application of Kimura-2 programme the present comparative study of nucleotide sequences revealed that maximum genetic divergence was between An. subpictus and An. annularis in which it gave a value of 1.384916 while it was minimum between An. culicifacies and An. stephensi with a value of 0.008244. The same was reflected in the phylogenetic trees generated by maximum parsimony (MP) method which showed that An. annularis and An. subpictus formed a single clade with a bootstrap value of 92.5 where An. stephensi also clustered with them with a bootstrap value of 99. Similarly, An. culicifacies with a bootstrap value of 100 also clustered with these species. When compared with the outgroup, An. maculatus closely paired with Cx. quinquefasciatus (Figure 2).
3.3 Sequence analysis of ITS2
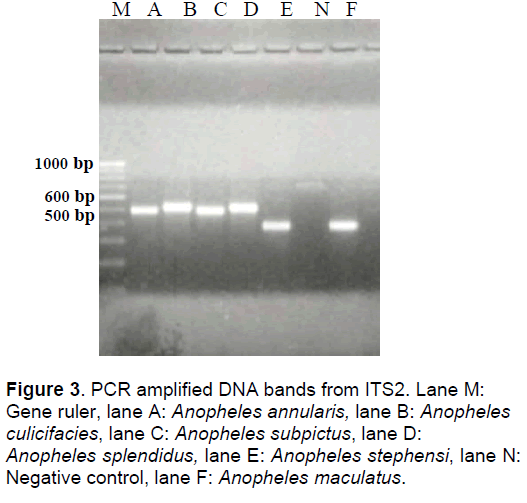
The PCR amplification of this spacer region produced a single DNA band from each species with a base pair length ranging from 458-506 bp. The sequence of An. stephensi had a minimum number of 458 bp followed by An. maculatus- 459 bp, An.annularis- 478 bp, An. splendidus- 488 bp, An. subpictus- 491, and An. culicifacies- 506 bp (Figure 3).
In majority of the anopheline species studied so far, the size of ITS2 region varies in length from 300-500 bp. For example, in the subgenus Cellia, An. gambiae has an ITS2 comprising of 426 bp, An. dirus complex has 710-716 bp while An. punctulatus has 549-563 bp. Within the subgenus Anopheles of the genus Anopheles, An. maculipennis has an average length of 305 bp, An. lesteri 438 bp, An. sinensis 459 bp, An. messeae 489 bp while An. quadrimaculatus complex has 305-310 bp. Similarly, within the subgenus Nyssorhynchus, An. nuneztovari has a sequence ranging from 363-369 bp while An. albitarsis has 490 bp.
As a result of these sequence comparisons, the average G:C percentage of this spacer region in the present species was higher than A:T as it was 55.34% in An. maculatus, 51.26% in An. annularis, 52.75% in An. subpictus, 55.90% in An. stephensi, 54.10% in An. splendidus and 60.28% in An. culicifacies. When compared with other anopheline species worked out so far, the G:C content was found to vary from 45.3% to 55.7% which falls well within the range of species under study. This increase in G:C has been attributed to evolutionary genetic drift of adaptive significance. With respect to substitutions, An. culicifacies had maximum number of 87 single nucleotide changes out of which there were 72 transversions and 15 transitions. The most frequent transversions were between adenine and thymine and, as compared to the incidence of substitution, the rate of transversions was found to be 66.78% which was more than the transitions which accounted for only 29.68%. As for the indels, there were a total of 274 loci with either the deletion or insertion of nucleotides. The maximum number of 59 indels was in An. annularis as compared to 54 in An. stephensi and An. maculatus, 50 in An. subpictus and An. splendidus and only 7 in An. culicifacies. With this, the transition to transversion ratio varied from 0.21 in An. culicifacies to 1 in An. splendidus. These values were considered significant, as substitutions are the main sources of evolution and speciation [12-14]. From the present results it is evident that, except for An. culicifacies, the ITS2 sequence of the remaining species is under rapid evolutionary changes due to its less conserved nature.
3.4 ITS2 sequence based phylogenetics
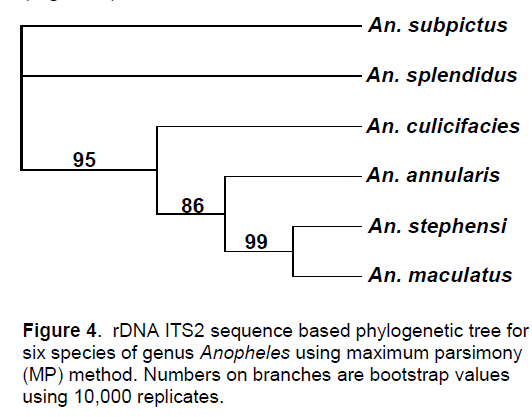
As per Kimura-2 parameter, there was maximum similarity between An. maculatus and An. stephensi which shared a value of 0.027620 while An. splendidus was found to be closest to An. subpictus with a value of 0.1127. The tree generated by MP method clearly indicated the close genetic homology and phylogenetic relationship of An. stephensi with An. maculatus due to their inclusion in a single clade with bootstrap values of 99 and 100 respectively (Figure 4).
For the remaining species, three different clades were generated in which one consisted of An. stephensi while the other had An. culicifacies included in it. In the same way, another clade included An. subpictus and An. splendidus. An. subpictus of Pyretophorus series showed a close relationship with An. splendidus of the series Neocellia. Since An. culicifacies is a member of Myzomyia series, it formed a separate clade with a bootstrap support value of 56%. In the overall assessment, it was found that maximum closeness was present between An. stephensi and An.maculatus while maximum divergence was between An. stephensi and An. subpictus.
3.5 Sequence analysis of COII gene
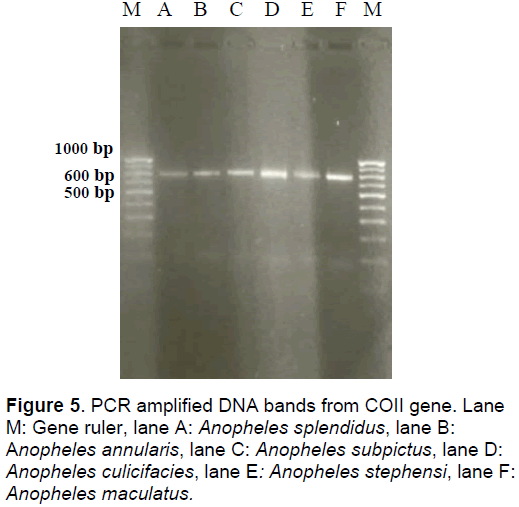
The amplified part of COII gene generated a band of 708-718 bp. The total sequence has ATG for initiation at 5’ end and only T at the 3’ end that codes for the entire set of termination sequence with 708 bp in An. maculatus, 710 bp in An. culicifacies, 711 bp each in An. stephensi and An. annularis, 713 in An. splendidus and 718 in An. subpictus (Figure 5).
Instead of G:C rich sequences of ITS1 and ITS2, the sequence of this gene was found to be A:T rich with maximum percentage of 75.07% in An. culicifacies followed by 73.87% in An. maculatus, 73.70% in An. stephensi, 72.70% in An. subpictus, 72.51% in An. splendidus and 71.45% in An. annularis. The A:T% of the sequences shows that out of all the species, An. culicifacies was closest to An. maculatus with a percentage values of 75.07% and 73.87% respectively. The sequence with 718bp was the longest in An. subpictus followed by 713bp in An. splendidus, 711bp in An. stephensi and An. annularis, 710 in An. culicifacies and smallest with 708bp in An. maculatus. The incidence of 35 transitions was maximum in An. maculatus followed by 22 in An. splendidus, 21 in An. annularis, 19 in An. stephensi and 17 each in An. subpictus and An. culicifacies. Similarly, transversions were also maximum with 44 in An. maculatus followed by 36 in An. splendidus, 26 in An. culicifacies, 25 in An. annularis, 24 in An. stephensi, and a minimum of 21 in An. subpictus. With this, the transition/ transversion ratio was found to range from 0.6 to 0.8.
In addition to these variations, indels were also observed at several places along the sequence. Out of a total of 66 loci where insertions or deletions had taken place, the minimum incidence of loss or gain of bases was found in An. subpictus while maximum of 14 in An. maculatus.
3.6 COII gene sequence based phylogenetics
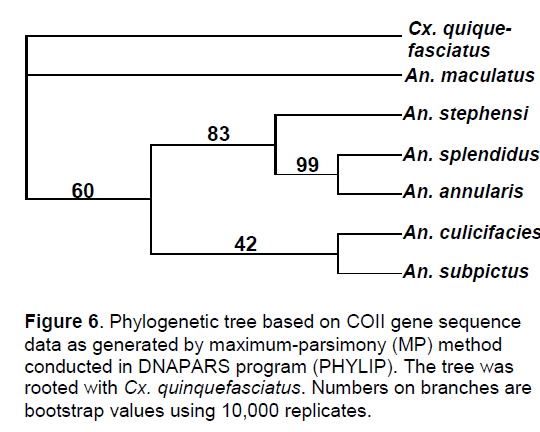
The phylogenetic trees showed the closeness of An. annularis and An. splendidus with bootstrap values of 100. According to the tree based on maximum parsimony (MP), An. stephensi formed a clade with An. annularis and An. splendidus with bootstrap value of 83 while An. culicifacies and An. subpictus formed another clade with a bootstrap value of 42 (Figure 6).
ITS1 and ITS2 are the noncoding sequences in the rDNA domain whereas COII gene sequence is a functional subunit of the mt.DNA. Therefore, on the basis of the studies carried out so far on the coding characteristics of this sequence in mosquitoes, it has been found that the whole sequence codes for 228-229 amino acids. For example, based on the coding characteristics An. stephensi and An. culicifacies once again made a distinct group. This is attributed to their anthropophilic and zoophilic feeding preferences which have given them the dubious status of urban and rural vectors respectively.
4. Discussions and Conclusions
In order to study the phylogeny of the six most important species of the genus Anopheles three molecular markers were used. Out of which ITS1 and ITS2 are highly variable regions of nuclear rDNA spacer sequences, while COII gene sequence is a highly conserved sequence of mitochondrial DNA. In addition to the ITS1, ITS2 and COII based assessment of genomic novelties in the present six species, the application of SpectraL Repeat Finder (SRF) programme also revealed valuable data on different types of repeats in the sequences. For example, polymers TGACCGA and CCTCGGC were remarkably similar in their copy number in An. stephensi and An. culicifacies. Documentation of such repeats may also be helpful in the selection of matching restriction enzymes in RFLP-PCR based studies of target species.
The genetic information about them is mainly based on polytene chromosome characteristics. With the advent of molecular parameters of study, a number of molecular markers are currently being used to characterize species with doubtful taxonomic status and phylogenetic relatedness. These molecular markers include noncoding sequences like ITS1 and 2 (noncoding) and coding sequences such as cytochrome b (cyt. b), cytochrome oxidase I and II (CO I, II), NADH dehydrogenase I, II, III, IV, V and VI. In order to augment the existing data, it would be appropriate to include more number of coding and noncoding sequences in the nuclear DNA and all the 37 mitochondrial genes.
The sequencing survey and phylogenetic analysis indicate greater diversity in the ITS1 sequence among the species covered in the present research programme. The efforts at exploiting the sequence variations of ITS2 region for species discrimination and phylogenetic analysis have proved quite useful in those cases where earlier parameters of study such as banding pattern of the polytene chromosomes and isozyme/allozyme variations were inconclusive[15,16].
The true function of these spacers remains vague, seemingly based on hydrogen-bonded secondary structure of RNA which, when modified slightly in conserved regions or modified considerably in variable regions, hinder maturation of the mRNA products [17]. Although, not as widely used as ITS2 in mosquitoes, ITS1 has similar properties to ITS2 and has been used at the population level in some insect groups [18]. In the Indian subcontinent genus Anopheles is represented by about 59 species out of which nearly 15 are confirmed vectors of malaria. The combined data sets of protein coding versus ribosomal genes will give a better understating of the different levels of systematic hierarchy and phylogenetic relationships.
Acknowledgements
The authors are thankful to Chairperson, Department of Zoology, Panjab University, Chandigarh for providing the necessary facilities under Centre of Advance Studies Programme of U.G.C, New Delhi, India to carry out the present research work.
References
- Marinucci M., Romi R., Mancini P., et al. (1999) Phylogenetic relationships of seven palearctic members of the maculipennis complex inferred from ITS2 sequence analysis. Insect Mol. Biol, 8(4): 469-480.
- Proft J., Maier W.A., Kampen H. (1999) Identification of six sibling species of the Anopheles maculipennis complex (Diptera: Culicidae) by a polymerase chain reaction assay. Parasitol Res, 85(10): 837-843.
- Wilkerson R.C., Parsons T.J., Klein T.A., et al. (1995) Diagnosis by random amplified polymorphic DNA polymerase chain reaction of four cryptic species related to Anopheles (Nyssorhynchus) albitarsis (Diptera: Culicidae) from Paraguay, Argentina, and Brazil. J Med Entomol, 32(5): 697-704.
- Chaudhry S., Neetu., Dhanda R.S., et al. (2004) Random amplified polymorphic DNA-polymerase chain reaction based differentiation of some species of the genus Anopheles (Culicidae: Diptera). J Cytol Genet, 5(NS): 173-183.
- Morgen J.A.T., Blair D. (1998) Relative merits of nuclear ribosomal internal transcribed spacers and mitochondrial COI and ND1 genes for distinguishing among Echinostoma species, Parasitology, 116: 289-297.
- Navajas M., Lagnel J., Gutierrez J., et al. (1998) Species-wise homogeneity of nuclear ribosomal ITS2 sequences in the spider mite Tetranchysis urticae contrasts with extensive mitochondrial COI polymorphism. Heredity, 80: 742-752.
- Marrelli M.T., Honorio N.A., Flores-Mendoza C., et al. (1999) Comparative susceptibility of two members of the Anopheles oswaldoi complex, An. oswaldoi and An. konderi, to infection by Plasmodium vivax. Trans R Soc Med Hyg, 93(4): 381-384.
- Hackett L.W., Missiroli A. (1935) The varieties of Anopheles maculipennis and their relation to the distribution of malaria in Europe. Riv Malariol, 14: 45-109.
- Hwang U-W., Kim W. (1999) General properties and phylogenetic utilities of nuclear ribosomal DNA and mitochondrial DNA commonly used in molecular systematics. Kor J Parasitol, 37(4): 215-228.
- Chaudhry S., Sharma M., Gupta S., et al. (2006) Multiple technique based species discrimination in the taxon Anopheles (Cellia) stephensi (Culicidae: Diptera). In: Vector Biology (eds. Sharma, V.P. and Kirti, J.S.). The National Academy of Sciences, India, pp. 105-123.
- Chaudhry S., Kohli, R. (2007) Sequence analysis of mitochondrial 16S ribosomal RNA gene fragment in two populations of Culex quinquefasciatus Say (Culicidae: Diptera). Natl Acad Sci Lett, 30(1&2): 55-60.
- Hillis D.M., Dixon M.T. (1991) Ribosomal DNA- molecular evolution and phylogenetic inference. Quarterly Rev Biol, 66: 410-453.
- Manonmani A.M., Townson H., Adeniran T., et al. (2001) rDNA-ITS2 polymerase chain reaction assay for the sibling species of Anopheles fluviatilis. Acta Trop, 78(1): 3-9.
- Porter C. H., Collins F. H. (1991) Species diagnostic differences in rDNA internal transcribed spacer from the sibling species Anopheles freeborni and Anopheles hermsi (Diptera: Culicidae). Am J Trop Med Hyg, 45(2): 271-279.
- Severini C., Silverstrini F., Mancini P., et al. (1996) Sequence and secondary structure of the rDNA second internal transcribed spacer in the sibling species Culex pipiens L. and Cx. quiquefasciatus Say (Diptera: Culicidae). Insect Mol Biol, 5(3): 181-186.
- Marrelli M.T., Floeter-Winter L.M., Malafronte R.S., et al. (2005) Analysis of the second internal transcribed spacers (ITS2) of ribosomal DNA sequences of the anopheline species from Amazon region, South America. Med Vet Entomol, 19: 208-217.
- van der Sande C.A.F.M., Kwa M., Van Nues R.W., et al. (1992) Functional analysis of internal transcribed spacer 2 of Saccharomyces cerevisiae ribosomal DNA. J Mol Biol, 223: 899-910.
- McLain D.K., Wesson D.M., Oliver J.H. Jr., et al. (1995) Evolution of the rDNA spacer, ITS2, in the ticks Ixodes scapularis and I. pacificus (Acari: Ixodi dae). Heredity, 75: 303-319.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences